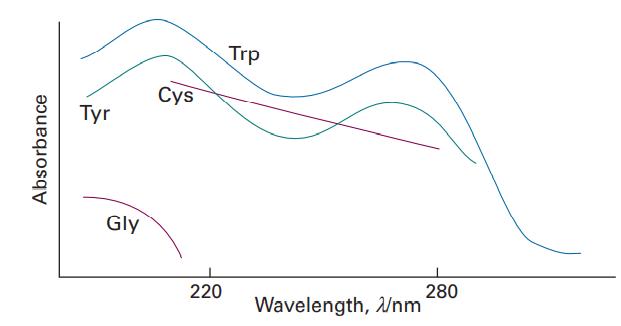
Figure 13.2 shows the UV-visible absorption spectra of a selection of amino acids. Suggest reasons for their
Question:
Figure 13.2 shows the UV-visible absorption spectra of a selection of amino acids. Suggest reasons for their different appearances in terms of the structures of the molecules.
Data in Figure 13.2
Transcribed Image Text:
Absorbance Tyr Gly Cys 220 Trp Wavelength, Wnm 280
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
Answer The different appearances of the UVvisible absorption spectra ...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
The 20 naturally occurring amino acids (See the following table) are all l amino acids, and they all have the S configuration, with the exception of glycine (which lacks a chirality center) and...
-
The UV spectrum of 1-phenylprop-2-en-1-ol shows an intense absorption at 220 nm and a weaker absorption at 258 nm. When this compound is treated with dilute sulfuric acid, it rearranges to an isomer...
-
A mixture of amino acids that do not separate sufficiently when a single technique is used can often be separated by two-dimensional chromatography. In this technique, the mixture of amino acids is...
-
aj b) If the magnitude of the force F is 700 N, what is the value of the internal moment at point A in N.m? d) 0.5 m 540 180 210 A 0.8 m 240 Bo birak F A
-
What do the terms debit and credit mean?
-
The following accounting issues have arisen at Tri-City Computers Corp.: 1. An investor noted that the market price of shares seemed to decline after the date of record for a cash dividend. Why do...
-
Oncology Tech is a manufacturer of cancer therapy devices called compensators. CMS was a company that sold cancer treatment planning software for use in purchasing cancer treatment devices. In 2006,...
-
1. Imagine that an agency or business has asked for help in gathering data about the number of sports anglers who fish off the coast of Georgia. What advice would you give about sampling? What method...
-
In return for an investment of $34318 in a fixed interest security, you will receive $134 at the end of each half year plus your money back on redemption in 12 years. You intend to deposit all the...
-
Jaylen has asked you to make a retirement planning presentation to the employees at Troy Metals Inc. as part of a financial literacy day. Your presentation will last approximately 60 minutes with...
-
3-Buten-2-one (3) has a strong absorption at 213nm and a weaker absorption at 320nm. Justify these features and assign the ultraviolet absorption transitions. 3 3-Buten-2-one
-
Estimate the magnitude of the transition dipole moment of a chargetransfer transition modelled as the migration of an electron from a H1s orbital on one atom to another H1s orbital on an atom a...
-
In the solution to the Blue Ridge Hot Tubs problem, the optimal values for X1 and X2 turned out to be integers (whole numbers). Is this a general property of the solutions to LP problems? In other...
-
Tony's Tables makes high-end, custom boardroom tables. The company applies overhead costs to jobs on the basis of direct labour hours. The company estimates manufacturing overhead for the year to be...
-
1. Given the following information about ABC corporation: Security Book Value Amount ($ millions) Common Stock Preferred Stock Bonds Retained Earnings Total 10 5 2 22 The common stock is currently...
-
On December 29, Larson sold products to a customer. As of December 31, Larson has not yet collected cash or billed the customer, but expects to receive cash in January. What are the effects of the...
-
Barry Computer Co. currently has $200,000 in AR with a DSO of 50 days. It wants to tighten its DSO to 30 days which it thinks will decrease sales by 10%. What will AR approximately equal following...
-
Determine the work done by the electric field of a 6 V battery on an electron traveling in a wire from the negative 0 V terminal to the positive 6 V terminal. Show your work below, paying close...
-
You are a publisher of national repute and you would like to find the potential for an academic book. You have decided to conduct a survey among college professors for this purpose. What form of...
-
In Exercises, find the equation of the tangent line at the given point on each curve. 2y 2 - x = 4; (16, 2)
-
Explain without using equations why (H /P)T 0H 0P is generally small for a real gas.
-
Why is it reasonable to write dH C P dT + VdP for a liquid or solid sample?
-
Refer to Figure 1.10 and explain why (U/V) T is generally small for a real gas. Figure 1.10 Ideal gas Real gas Tranlaition rv-0 (4)A
-
SM Partners, LLP has taken on a new client during 2023, Perfect Polaroid, Inc., an issuer. PPI was audited by GMPK, a large public accounting firm, from 2018-2022 but chose to switch auditors in the...
-
Fast Deliveries, Incorporated (FDI), was organized in December last year and had limited activity last year. The resulting balance sheet at the beginning of the current year is provided below: FAST...
-
"Ecologically sustainable development is not perfect, however it is the best option we have" critically analysis this statement with reference to Australian State and Commonwealth Environmental Law

Study smarter with the SolutionInn App


