In an experiment on the Pt|H 2 |H + electrode in dilute H 2 SO 4 the
Question:
In an experiment on the Pt|H2|H+ electrode in dilute H2SO4 the following current densities were observed at 25°C. Evaluate α and j0 for the electrode.
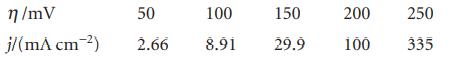
How would the current density at this electrode depend on the overpotential of the same set of magnitudes but of opposite sign?
Transcribed Image Text:
η/mV j/(m^ cm-²) 50 100 150 2.66 8.91 29.9 200 100 250 335
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 16% (6 reviews)
Solution Using the equations for PtH2H and PtH2H in dilute H2SO4 we have Now b...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In an experiment on the Pt IH,I H+ electrode in dilute H,S04 the following current densities were observed at 25e. Evaluate ET and jo for the electrode. How would the current density at this...
-
The following data were observed in an experiment on the photoelectric effect from potassium: Graphically evaluate these data to obtain values for the work function and Plancks constant. 1019 Kinetic...
-
In an experiment on soybean varieties, individually potted soybean plants were grown in a greenhouse, with 10 plants of each variety used in the experiment. From the harvest of each plant, five seeds...
-
Differentiate implicitly to find 2x + 5xy + 5y +16y - 8 = 0; dy dx . Then find the slope of the curve at the given point. (-2,0)
-
The data in the file named Fast100 were collected by D. L. Green & Associates, a regional investment management company that specializes in working with clients who wish to invest in smaller...
-
(EPS: Preferred Dividends, Options, and Convertible Debt) Earnings per share (EPS) is the most featured, single financial statistic about modern corporations. Daily published quotations of stock...
-
Three bars, each made of different materials, are connected together and placed between two walls when the temperature is T 1 = 12C. Determine the force exerted on the (rigid) supports when the...
-
Create class diagrams that describe the classes and relationships depicted in the following scenarios: Researchers are placed into a database that is maintained by the state of Georgia. Information...
-
(d) Write a PHP script to find the first non-repeated character in a given string. Example #1 Sample Input: apple Sample Output: a Example #2 Sample Input: abcab Sample Output: c Example #3 Sample...
-
Foreign Exchange Trading at Baldwin Enterprises Baldwin Enterprises is a large manufacturing company with operations and sales divi- sions located in the United States and several other countries....
-
In an experiment on the adsorption of oxygen on tungsten it was found that the same volume of oxygen was desorbed in 27 min at 1856 K and 2.0 min at 1978 K. What is the activation energy of...
-
Nitrogen gas adsorbed on charcoal to the extent of 0.921 cm 3 g 1 at 490 kPa and 190 K, but at 250 K the same amount of adsorption was achieved only when the pressure was increased to 3.2 MPa. What...
-
For amounts between $10,000 and $24,999, a credit union pays a rate of 0.5% on term deposits with maturities in the 91-to-120-day range. However, early redemption will result in a rate of 0.2% being...
-
Larry purchased an annuity from an insurance company that promises to pay him $500 per month for the rest of his life. Larry paid $48,180 for the annuity. Larry is in good health and is 72 years old....
-
Consider a multivariate linear regression problem of mapping Rd to R, with two different objective functions. The first objective function is the sum of squared errors, as presented in class; i.e.,...
-
Suppose that you have an iid sample: (Wn, Fn)-1 where Wn is person n's hourly wage and F is a binary variable equal to 1 if person n is female and 0 other- wise. Consider the linear model E[log(Wn)...
-
How much money should you save per year if you want to have $ 1,000,000 in 20 years. Corresponding interest rate is 13%? Answer:
-
Use the exhibits in the Excel file to prepare a six-month budget. Use the blanks tab in the Excel file to prepare: a. Sales budget b. Production budget (note: ending inventory should be 300 units 20%...
-
What is the relationship between vitamins and coenzymes?
-
In the figure, two loudspeakers, separated by a distance of d1 = 2.63 m, are in phase. Assume the amplitudes of the sound from the speakers are approximately the same at the position of a listener,...
-
One mole of H 2 O(l) is super cooled to 3.75C at 1 bar pressure. The freezing temperature of water at this pressure is 0.00C. The transformation H 2 O(l) H 2 O(s) is suddenly observed to occur. By...
-
An athlete at high performance inhales ~3.75 L of air at 1.0 atm and 298 K at a respiration rate of 32 breaths per minute. If the exhaled and inhaled air contain 15.3 and 20.9% by volume of oxygen,...
-
The temperature of 1.75 moles of an ideal gas increases from 10.2C to 48.6C as the gas is compressed adiabatically. Calculate q, w, U, and H for this process assuming that C V ,m = 3/2 R.
-
The XYZ Company is considering two alternative methods of producing washing powder. Under the method A the fixed cost would be $40,000 and the variable cost per unit $6.60, while under the method B...
-
Esquire Company needs to acquire a molding machine to be used in its manufacturing process. Two types of machines that would be appropriate are presently on the market. The company has determined the...
-
Stillicum Corporation makes ultra light-weight backpacking tents. Data concerning the company's two product lines appear below: Direct materials per unit Direct labor per unit Direct labor-hours per...

Study smarter with the SolutionInn App


