The chlorination of vinyl chloride, C 2 H 3 Cl + Cl 2 C 2 H 3
Question:
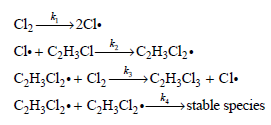
Derive the rate law expression for the chlorination of vinyl chloride based on this mechanism.
Transcribed Image Text:
Cl2-2C1. Cl. + C2H3CI- С Н.С1,-+ Cl, — сн,C + —Сн.Cl. Cl. →stable species СН.С1,.+ Снс1,.—
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
The rate of vinyl chloride production is The rate ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The reaction I2(aq) + OCl2(aq) IO2(aq) + Cl2(aq) is believed to occur by the following mechanism: Write the rate law for this reaction.
-
The hydrogenbromine reaction corresponds to the production of HBr(g) from H 2 (g) and Br 2 (g) as follows: H 2 (g) + Br 2 (g) 2HBr(g). This reaction is famous for its complex rate law, determined by...
-
For the reaction I (aq) + OCl (aq) OI (aq) + Cl (aq) occurring in aqueous solution, the following mechanism has been proposed: a. Derive the rate law expression for this reaction based on this...
-
Calculate the following integral approximately by using the Simpson's method: 3.6 f. f(x) dx X 0.0 0.6 1.2 1.8 2.4 3.0 3.6 f(x) 0 0.13 0.05 0.6 0.43 0.74 0.93
-
The function f(x) = k(2 x x3) has an inverse function, and f1(3) = 2. Find k.
-
What is your strategic focus for early growth? How do you leverage what you do really well? What are some possible peripheral growth opportunities for later in your ventures life?
-
In refrigeration cycle, heat is (a) Abstracted from the lower temperature region and discarded to the higher one (b) Absorbed from the higher temperature region and discarded to the lower one (c)...
-
City Sights, Ltd., operates a tour and sightseeing business. Its trademark is the use of trolley buses. Each vehicle has its own identity and is specially made for the company. Gridlock, the oldest...
-
What reform measures have been instituted in the state of Tennessee in regards to damage caps, joint and several liability, the collateral-source rule, and frivolous lawsuits? How effective have they...
-
On 1 April, 2019 IKe purchased 80% of equity shares in Rosemary. On the same date IKe acquired 40% of the 40 million equity shares in Abena paying Ghs2 per share. The statement of profit or loss for...
-
Consider the collision-induced dissociation of N2O5(g) via the following mechanism: The asterisk in the first reaction indicates that the reactant is activated through collision. Experimentally it is...
-
In the unimolecular isomerization of cyclobutane to butylene, the following values for kuni as a function of pressure were measured: Assuming that the Lindemann mechanism accurately describes this...
-
Daytona Wheels is a Japanese tire manufacturer. For August 2021, it budgeted to manufacture and sell 3,000 tires at a variable cost of $74 per tire and total fixed costs of $54,000. The budgeted...
-
Bolsters food balance sheet shows a total of 25 million long term debt with a coupon rate of 8.50 the yield to maturity on this debt is 8.00 and the debt has a total current market value of 27...
-
Consider an 11-year, corporate bond with face value $1,000 that pays semi-annual coupon. With the nominal yield-to-maturity equal to 10%, the bond is selling at $802.5550. Find the coupon rate for...
-
An insurance company must make payments to a customer of $1 million in one year and $400,000 in four years. The yield curve is flat at 10%. Use annual compounding. If the company wants to fully fund...
-
4. Consider a European put option on a stock. The stock price is $70, the time to maturity is 8 months, the risk-free rate of interest is 10% per annum, the exercise price is $65, and the volatility...
-
Kentville Petroleum needs $1,000,000 to take advantage of a cash discount of 2/20, net 60. A banker will loan the money for 40 days at a total interest cost of $10,959 a) What is the annual rate on...
-
A license plate consists of 1 letter, excluding O and I, followed by a 4-digit number that cannot have a 0 in the lead position. How many different plates are possible?
-
Evaluate how many lines there are in a true rotational spectrum of CO molecules whose natural vibration frequency is w = 4.09 1014 s1 and moment of inertia I = 1.44 1039 g cm2.
-
Identify the missing reagent needed to achieve the following transformations: a. b. c. d. Br KF benzene Br NaF benzene
-
Consider the oxidation of the amino acid glycine NH 2 CH 2 COOH to produce water, carbon dioxide, and urea NH 2 CONH 2 : NH 2 CH 2 COOH(s) + 3O 2 (g) NH 2 CONH 2 (s) + 3CO 2 (g) + 3H 2 O(l)...
-
Show what reagents you would use to prepare each of the following ethers via a Williamson ether synthesis, and explain your reasoning. a. b. c. OMe
-
Balance per bank statement Less: outstanding cheques ARKIN Company Bank reconciliation May 31, 2014 6124 6138 6139 6140 add: deposit in transit 2125 932,65 30928,46 960,57 1420 5438,22 25490,24...
-
Company "A" is a manufacturer of wooden and metal structures (20XX) Costs 1. Consumable supplies in the factory warehouse 2. Sandpaper 3. Lubricants 4. Indirect costs: wages 5. Wages of production...
-
Write down at leastfive items (durable goods, not food) that you purchase and their sourcing (where each is from). For example, a shirt may be assembled in China, designed in the US, and made from...

Study smarter with the SolutionInn App


