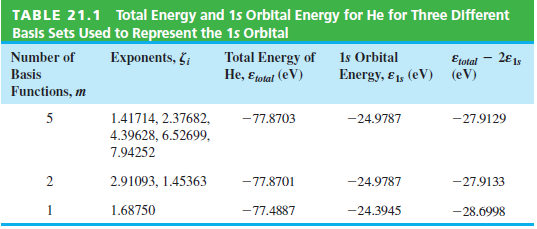
The exact energy of a ground-state He atom is 79.01 eV. Calculate the correlation energy and the
Question:
Transcribed Image Text:
TABLE 21.1 Total Energy and 1s Orbital Energy for He for Three Different Basis Sets Used to Represent the 15 Orbital Number of Exponents, §; Total Energy of He, Eotal (eV) Is Orbital Etotal – 2€1, (eV) Basis Energy, £1, (eV) Functions, m 1.41714, 2.37682, 4.39628, 6.52699, 5 -77.8703 -27.9129 -24.9787 7.94252 2.91093, 1.45363 -77.8701 -24.9787 -27.9133 1 1.68750 -77.4887 -24.3945 -28.6998
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
The correlation energy is the difference between the exact ...View the full answer

Answered By

Dinesh F
I have over 3 years of professional experience as an assignment tutor, and 1 year as a tutor trainee.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The binding energy of an electron in the ground state of He atom is equal to E o - 24.6 eV. Find the energy required to remove both electrons from the atom.
-
A hydrogen atom is excited from its ground state to the state with n = 4. (a) How much energy must be absorbed by the atom? Consider the photon energies that can be emitted by the atom as it...
-
(Multiple choice) (1) The energy of the ground state of doubly ionized lithium (Z = 3) is ______, where E 0 = 13.6 eV. (a) 9E 0 , (b) 3E 0 , (c) E 0 /3, (d) E 0 /9. (2) Bohrs quantum condition on...
-
Compensation System Change Introduction As Melanie Griffith gazed through the window of her office, she could see some employees walking to the parking lot to get to their cars; others were on their...
-
A ball that is bobbing up and down on the end of a spring has a maximum displacement of 3 inches. Its motion (in ideal conditions) is modeled by y = 1/4 cos 16t, t > 0, where y is measured in feet...
-
What is needed in order to deduct a business expense on a tax return?
-
Why are information security and privacy important considerations in the design, development, and maintenance of an HRIS?
-
The Spring family has owned and operated a garden tool and implements manufacturing company since 1952. The company sells garden tools to distributors and also directly to hardware stores and home...
-
Sage Hill Company has been in business several years. At the end of the current year, the unadjusted balances show: Accounts Receivable Sales Revenue Allowance for Doubtful Accounts $272,800...
-
21. A shaft made of 40 C 8 steel is used to drive a machine. It rotates at 1500 rp.m. The pulleys.A, B and the bearings C, D are located as shown in Fig. 14.21. The belt tensions are also shown in...
-
The ground-state wave function of Li 2+ is -1/2 (Z/a 0 ) 3/2 e -Zr /a 0 , where Z is the nuclear charge. Calculate the expectation value of the potential energy for Li 2+ .
-
Write the Slater determinant for the ground-state configuration of Be.
-
Why is activity analysis important?
-
In what ways are we all farmers?
-
Health disparities are found not only across different countries but also within countries. Ask students to share health disparities that they are aware of in the United States. Choose one or two...
-
What is the difference between GDP and NNW?
-
What do you think is more Chinese? A German person using acupuncture to treat lower back pain or a Chinese person using surgery to relieve back pain? What cultural issues are involved in both of...
-
In your own words thoroughly explain the identification problem and give an example.
-
Suppose that (a) What is F(-5)? What point is on the graph of F? (b) If F(x) = 24, what is x? What point is on the graph of F? (c) Find the zero of F. - 3. F(x)
-
Q1) What is the a3 Value Q2) What is the a7 Value Q3) What is the a4 Value Q4) What is the b3 Value Q5) What is the b2 Value Q6) What is the sign of 2nd constraint? A pastry chef at a bakery wants to...
-
Explain the origin of colligative properties.
-
How is Raoults law modified so as to describe the vapour pressure of real solutions?
-
At 300 K, the partial vapour pressures of HCl (that is, the partial pressure of the HCl vapour) in liquid GeCl 4 are as follows: Show that the solution obeys Henrys law in this range of mole...
-
Create a segmentation, selection and focus model that allows the PUMA company in Mexico to determine its customer, where to find it and how to generate value given its model. 7) Selection of the most...
-
Determine if the following values are zeroes polynomial. 4 3 f(x) = x + x-18x - 16x + 32 x+1, +2, +4 -
-
Cali Windows is a small company that installs windows. Its cost structure is as follows: (Click the icon to view the cost structure.) Calculate (a) the breakeven point in units and revenues and (b)...

Study smarter with the SolutionInn App


