FIGURE EX38.24 is an energy-level diagram for a simple atom. What wavelengths, in nm, appear in the
Question:
FIGURE EX38.24 is an energy-level diagram for a simple atom. What wavelengths, in nm, appear in the atom??s
(a) Emission spectrum
(b) Absorption spectrum?
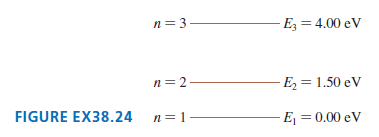
Transcribed Image Text:
n= 3 Ez = 4.00 eV n=2- E = 1.50 eV FIGURE EX38.24 n= 1 E = 0.00 eV
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
Model To conserve energy the emission and the absorption photons ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Figure 29.7 is an energy level diagram for 208Tl. What are the energies of the photons emitted for the six transitions shown? Figure 29.7 492 keV 472 keV 327 keV 40 keV
-
Show an energy level diagram for the MOs for He 2 and show how the electrons would be arranged in these MOs.
-
Draw an energy level diagram for the excited state of H2. Is there still a bond between the hydrogens?
-
A mail-order firm processes 5,300 checks per month. Of these, 60 percent are for $55 and 40 percent are for $80. The $55 checks are delayed two days on average; the $80 checks are delayed three days...
-
How does E = mc 2 describe the identities of energy and mass?
-
Row-reduce the matrix without the aid of a calculator, indicating the row operations you are using at each step using the notation of Definition RO. 1. 2. [21510 1-3-1 -2 4 -2 6 12 12-4
-
Suppose that a continuous-time compounding framework is used with a fixed interest rate \(r\). Suppose that the carrying charge per unit of time is proportional to the spot price; that is, the charge...
-
Jefferson Products, Inc., is considering purchasing a new automatic press brake, which costs $300,000 including installation and shipping. The machine is expected to generate net cash inflows of...
-
On January 2, 2018, Uhrhan, Inc., signed an eight-year lease for office space. Uhrhan classified the lease as a finance lease. Uhrhan has the option to renew the lease for an additional four-year...
-
The position of a particle as a function of time is given by r(vector) = (5.0i + 4.0j)t 2 m, where t is in seconds. a. What is the particles distance from the origin at t = 0, 2, and 5 s? b. Find an...
-
An electron with 2.00 eV of kinetic energy collides with the atom shown in FIGURE EX38.24. a. Is the electron able to excite the atom? Why or why not?b. If your answer to part a was yes, what is the...
-
What is the length of a one-dimensional box in which an electron in the n = 1 state has the same energy as a photon with a wavelength of 600 nm?
-
For the following exercises, use the descriptions of the pairs of lines to find the slopes of Line 1 and Line 2. Is each pair of lines parallel, perpendicular, or neither? Line 1: Passes through (5,...
-
Use the following information to answer question 3 and 4 Suppose that the current spot exchange rate is 0.80/$ and the bank quoted forward exchange rate is 0.7813/$. The one-year interest rate is...
-
A restriction enzyme is supplied at a concentration of 10 units/uL. If you use 2 L of the enzyme in a digestion reaction, how many units of the enzyme are you adding?
-
What is the present value of $3,525 per year, at a discount rate of 10 percent, if the first payment is received 7 years from now and the last payment is received 25 years from now? (Do not round...
-
A stock will provide a rate of return of either 25% or 38%. If both possibilities are equally likely, calculate the stock's expected return and standard deviation. (Do not round intermediate...
-
Oil Wells offers 5.6 percent coupon bonds with annual payments and a yield to maturity of 6.94 percent. The bonds mature in seven years. The face value is $1,000. What is the current yield?
-
The wavelength 10.0 m is in the infrared region of the electromagnetic spectrum, whereas 600 nm is in the visible region and 100 nm is in the ultraviolet. What is the temperature of an ideal...
-
Don Griffin worked as an accountant at a local accounting firm for five years after graduating from university. Recently, he opened his own accounting practice, which he operates as a corporation....
-
A countercurrent gas absorption tower packed with 1.5 in ceramic Intalox saddles is used to process 360 kgmole/h of an inlet gas stream containing 10 mole% sulfur dioxide (SO 2 ) in nitrogen (N 2 )...
-
A counter-current packed tower will be used to remove H 2 S vapor from a N 2 -rich gas stream. In the present process, the inlet gas stream entering the bottom of the tower contains 5.0 mole% H 2 S...
-
Consider the concentric tubular mass transfer device shown in the figure below. This device is designed to gradually introduce hydrogen (H 2 ) gas (species A) into a pure oxygen gas (O 2 ) inlet...
-
Find the integrating factor for the given 1-order linear non-homogeneous ordinary differential equation. Do not solve the ordinary differential equation. y xdx-xdy-x+dx + dx
-
[Bush] Consider the following snippet of code. (Assume that input strings, including null terminator, will always fit within the size 255 array.) char* to_upper_case(char* original) { char...
-
50. Show that if f(x) = a,x" + a-1x+...+x+ ao, a,..., a-1, and a,, are real numbers and where 0, then f(x) is O(x"). an # Big-O, big-Theta, and big-Omega notation can be extended to functions in more...

Study smarter with the SolutionInn App


