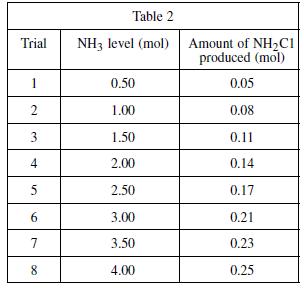
If a ninth trial were conducted in Experiment 3, adding 1.25 mol of NH 3 to the
Question:
If a ninth trial were conducted in Experiment 3, adding 1.25 mol of NH3 to the bleach–water solution, the amount of NH2Cl produced would be closest to:
A. 0.06 mol.
B. 0.10 mol.
C. 0.12 mol.
D. 0.16 mol.
Experiment 3
In yet another reaction, bleach and ammonia combined under certain conditions produce a compound known as chloramine. Chloramine (NH2Cl) is a toxic substance commonly used in low concentrations as a disinfectant in municipal water systems as an alternative to chlorination. To determine the mixture of bleach and ammonia at which NH2Cl is produced, a varying amount of ammonia was added to eight different bleach–water solutions and the resulting chlorine gas from each mixture was collected and measured. A solution of 1.0 mole (mol) of NaOCl in 1 kg of water was used in each trial. A certain quantity of NH3 was added to each solution; the quantity of ammonia added was gradually increased for each trial. The amount of chloramine produced in each trial was recorded in Table 2.
Step by Step Answer:






