Question: Based on Figure 21-6, what are the processes involved in atomic absorption of a solution of a metal ion? In atomic emission? Figure 21-6 Excitation
Based on Figure 21-6, what are the processes involved in atomic absorption of a solution of a metal ion? In atomic emission?
Figure 21-6
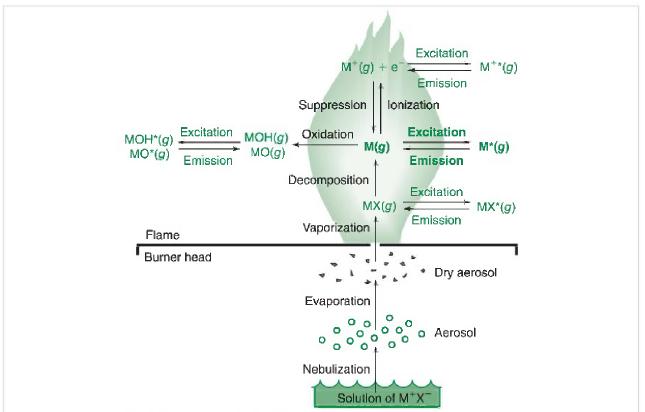
Excitation MOH(g) MO(g) MOH*(g) MO*(g) Emission Flame Burner head M*(g) + e Suppression Oxidation M(g) Decomposition MX(g) Vaporization Evaporation lonization Nebulization Excitation Emission Excitation Emission Excitation Emission Solution of M*X M**(g) M*(g) MX*(g) Aerosol Dry aerosol
Step by Step Solution
3.40 Rating (159 Votes )
There are 3 Steps involved in it
Solution The involved processes in atomic absorption of a solution of a metal ion are 1 In the absor... View full answer

Get step-by-step solutions from verified subject matter experts


