What color do you expect to observe for cresol purple indicator (Table 10-3) at the following pH
Question:
What color do you expect to observe for cresol purple indicator (Table 10-3) at the following pH values?
(a) 1.0;
(b) 2.0;
(c) 3.0
Table 10-3
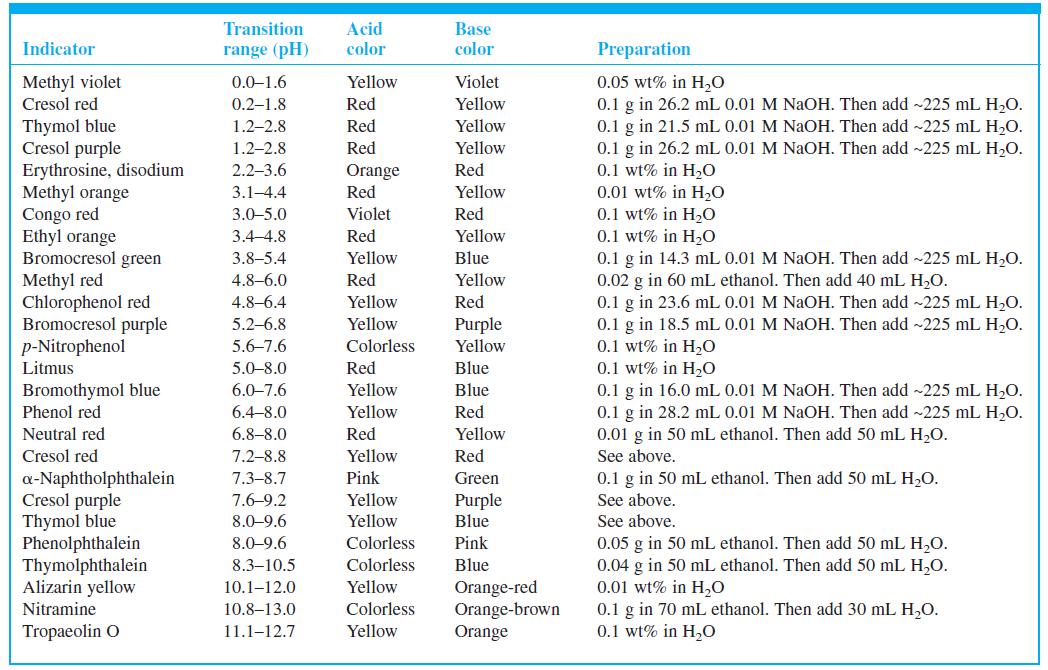
Acid color Transition Base Indicator range (pH) color Preparation Methyl violet Cresol red Violet 0.05 wt% in H,O 0.1 g in 26.2 mL 0.01 M NAOH. Then add ~225 mL H,O. 0.1 g in 21.5 mL 0.01 M NAOH. Then add -225 mL H,O. 0.1 g in 26.2 mL 0.01 M NaOH. Then add ~225 mL H,O. 0.1 wt% in H20 0.01 wt% in H2O 0.1 wt% in H0 0.1 wt% in H20 0.1 g in 14.3 mL 0.01 M NaOH. Then add ~225 mL H2O. 0.02 g in 60 mL ethanol. Then add 40 mL H,O. 0.1 g in 23.6 mL 0.01 M NaOH. Then add -225 mL H,O. 0.1 g in 18.5 mL 0.01 M NaOH. Then add ~225 mL H,O. 0.1 wt% in H20 0.1 wt% in H20 0.1 g in 16.0 mL 0.01 M NAOH. Then add ~225 mL H,O. 0.1 g in 28.2 mL 0.01 M NaOH. Then add ~225 mL H2O. 0.01 g in 50 mL ethanol. Then add 50 mL H,O. 0.0-1.6 Yellow 0.2-1.8 Red Yellow Thymol blue Cresol purple Erythrosine, disodium Methyl orange Congo red Ethyl orange Bromocresol green Methyl red Chlorophenol red Bromocresol purple p-Nitrophenol Litmus 1.2–2.8 Red Yellow 1.2-2.8 Red Yellow 2.2–3.6 Orange Red Red 3.1-4.4 Yellow 3.0-5.0 Violet Red 3.4-4.8 Red Yellow 3.8–5.4 Yellow Blue 4.8–6.0 Red Yellow 4.8–6.4 Yellow Red 5.2-6.8 Yellow Purple Yellow 5.6–7.6 Colorless 5.0-8.0 Red Blue Bromothymol blue Phenol red 6.0-7.6 Yellow Blue 6.4-8.0 Yellow Red Neutral red 6.8–8.0 Red Yellow Cresol red 7.2-8.8 Yellow Red See above. a-Naphtholphthalein Cresol purple Thymol blue Phenolphthalein Thymolphthalein Alizarin yellow 7.3–8.7 Pink Green 0.1 g in 50 mL ethanol. Then add 50 mL H2O. 7.6-9.2 Yellow Purple See above. 8.0-9.6 Yellow Blue See above. 8.0-9.6 Colorless Pink 0.05 g in 50 mL ethanol. Then add 50 mL H,O. 0.04 g in 50 mL ethanol. Then add 50 mL H,O. 0.01 wt% in H,0 0.1 g in 70 mL ethanol. Then add 30 mL H,O. 0.1 wt% in H0 8.3–10.5 Colorless Blue Yellow Orange-red Orange-brown Orange 10.1–12.0 Nitramine 10.8–13.0 Colorless Tropaeolin O 11.1–12.7 Yellow
Step by Step Answer:
From table given in question T...View the full answer



Related Video
A substance that alters color in solution over a constrained range of pH values is known as a pH indicator or acid-base indicator. The indicator chemical just needs to alter color slightly in order to be noticed. Indicators work on the basis that they react with water to produce the hydrogen cation H+ or hydronium ion H3O+. The indicator molecule\\\'s color changes as a result of the reaction. Since indicators have distinct color ranges for color change, they can occasionally be combined to provide color changes over a wider pH range.
Students also viewed these Engineering questions
-
Do you expect to observe more or less voluntary disclosure by companies in emerging-market countries than in developed countries? Why? Do you expect to observe more or less regulatory disclosure...
-
What type of compensation policy would you expect to observe in an industry with rapidly changing technology? Explain
-
Do you expect to see larger solidification shrinkage for a material with a bcc crystal structure or fee? Explain.
-
College Spirit sells sportswear with logos of major universities. At the end of 2019, the following balance sheet account balances were available. Accounts payable $104,700 Required: 1. Prepare a...
-
How can you determine when you are involved in tax planning? What is your role as a tax planner? How does this differ from tax research in which no planning is requested? Why is it important to...
-
Write an HDL module for the daughter snail from Exercise 3.25. Data from problem 3.25 Alyssa P. Hackers snail has a daughter with a Mealy machine FSM brain. The daughter snail smiles whenever she...
-
What is the future equivalent of \($1,000\) invested at 8% simple interest for 3 years?
-
In what ways might a rms suppliers improve or undermine the rms Lean efforts? Can you think of any examples from the chapter that illustrate this idea?
-
What would be the annual percentage yield for a savings account that earned $54 in interest on $750 over the past 365 days?
-
Carrie DLake, Reed A. Green, and Doug A. Divot share a passion for golf and decide to go into the golf club manufacturing business together. On January 2, 2020, DLake, Green, and Divot form the...
-
Consider the titration in Figure 10-2, for which the pH at the equivalence point is calculated to be 9.25. If thymol blue is used as an indicator, what color will be observed through most of the...
-
What color do you expect to observe for cresol purple indicator (Table 10-3) at the following pH values? (a) 1.0; (b) 2.0; (c) 3.0 Table 10-3 Acid color Transition Base Indicator range (pH) color...
-
The equations are identities because they are true for all real numbers. Use properties of logarithms to simplify the expression on the left side of the equation so that it equals the expression on...
-
Compute the primary and secondary Class 1 NICs payable in relation to the following employees: (a) A earns 198 for the week ending 25 June 2021. (b) B earns 803 for the week ending 25 June 2021. (c)...
-
How would the following two problems be resolved in your state? You may have to check both statutory law and case law. a. Fred and Jill are married. Fred is killed in an automobile accident with the...
-
Wardle, Jingle and Trotter began trading on 1 October 2019, preparing accounts to 30 September each year and sharing profits in the ratio 7:2:1. Results for the first two years of trading are as...
-
Frances paid income tax and Class 4 NICs of 47,000 for tax year 2020-21, of which 19,000 was paid via the PAYE system. Her total liability for 2021-22 is 69,000, of which 21,000 is paid via PAYE. Her...
-
Rose is single and has total income of 12,300 in 2021-22. She makes a gross deductible payment of 25 during the year. Show her income tax computation for the year and determine the amount of tax...
-
On December 31, 2016, Jabot purchased $16,000 of equipment on a one year, 9 percent note payable. Journalize the company's purchase of equipment, the accrual of interest expense on May 31, 2017 (its...
-
Doorharmony Company makes doorbells. It has a weighted- average cost of capital of 5% and total assets of $ 5,900,000. Doorharmony has current liabilities of $ 750,000. Its operating income for the...
-
A low-altitude meteorological research balloon, temperature sensor, and radio transmitter together weigh 2.5 lb. When inflated with helium, the balloon is spherical with a diameter of 4 ft. The...
-
A submarine releases a spherical flotation buoy containing a radio beacon. The buoy has a diameter of 1 ft and weighs 22 lb. The coefficient of drag for the submerged buoy is C D = 0.45. At what...
-
Place Equation (6.16) into the form of Equation (6.14) and show that the coefficient of drag for small Reynolds numbers is given by Equation (6.15). Equation 6.14 Equation 6.15 Equation 6.16 FD =...
-
ABC Inc. curently has a share price of $44. If they are paying a dividend of $1.13, with a cum dividend date of November 1, would would you expect their share price to be on November 2nd? Please give...
-
Does China benefit from a weak currency?Does Us benefit from a strong currency? Why? Please explain
-
You purchased 10 ASD June 45 puts and the company declares a 10% stock dividend. How are the terms of the option contract then changed?

Study smarter with the SolutionInn App


