What color do you expect to observe for cresol purple indicator (Table 10-3) at the following pH
Question:
What color do you expect to observe for cresol purple indicator (Table 10-3) at the following pH values?
(a) 1.0;
(b) 2.0;
(c) 3.0
Table 10-3
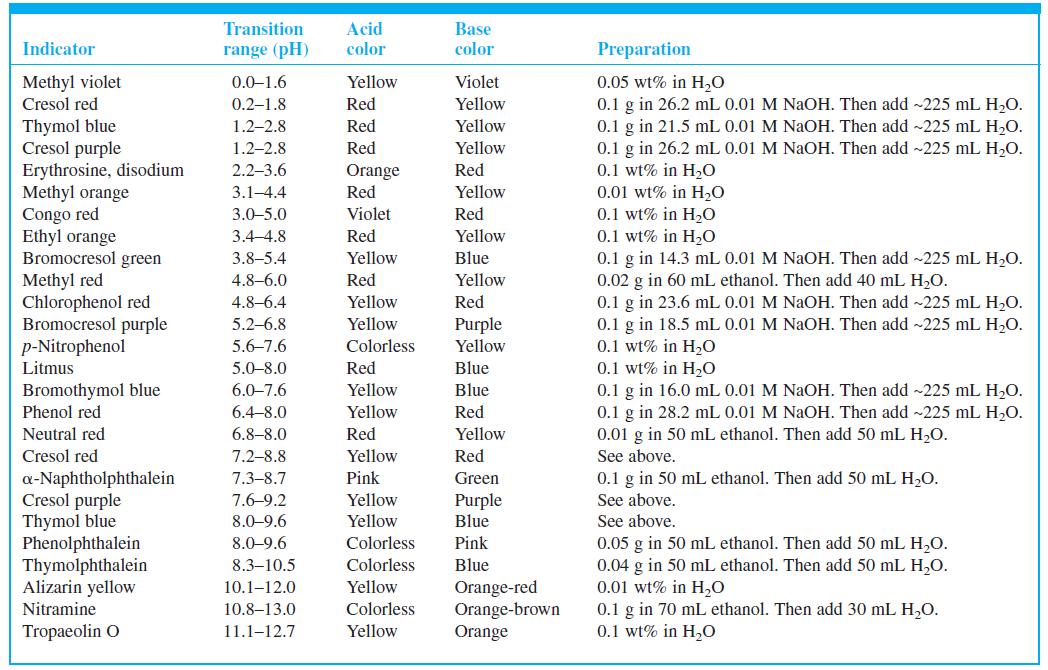
Acid color Transition Base Indicator range (pH) color Preparation Methyl violet Cresol red Violet 0.05 wt% in H,O 0.1 g in 26.2 mL 0.01 M NAOH. Then add ~225 mL H,O. 0.1 g in 21.5 mL 0.01 M NAOH. Then add -225 mL H,O. 0.1 g in 26.2 mL 0.01 M NaOH. Then add ~225 mL H,O. 0.1 wt% in H20 0.01 wt% in H2O 0.1 wt% in H0 0.1 wt% in H20 0.1 g in 14.3 mL 0.01 M NaOH. Then add ~225 mL H2O. 0.02 g in 60 mL ethanol. Then add 40 mL H,O. 0.1 g in 23.6 mL 0.01 M NaOH. Then add -225 mL H,O. 0.1 g in 18.5 mL 0.01 M NaOH. Then add ~225 mL H,O. 0.1 wt% in H20 0.1 wt% in H20 0.1 g in 16.0 mL 0.01 M NAOH. Then add ~225 mL H,O. 0.1 g in 28.2 mL 0.01 M NaOH. Then add ~225 mL H2O. 0.01 g in 50 mL ethanol. Then add 50 mL H,O. 0.0-1.6 Yellow 0.2-1.8 Red Yellow Thymol blue Cresol purple Erythrosine, disodium Methyl orange Congo red Ethyl orange Bromocresol green Methyl red Chlorophenol red Bromocresol purple p-Nitrophenol Litmus 1.2–2.8 Red Yellow 1.2-2.8 Red Yellow 2.2–3.6 Orange Red Red 3.1-4.4 Yellow 3.0-5.0 Violet Red 3.4-4.8 Red Yellow 3.8–5.4 Yellow Blue 4.8–6.0 Red Yellow 4.8–6.4 Yellow Red 5.2-6.8 Yellow Purple Yellow 5.6–7.6 Colorless 5.0-8.0 Red Blue Bromothymol blue Phenol red 6.0-7.6 Yellow Blue 6.4-8.0 Yellow Red Neutral red 6.8–8.0 Red Yellow Cresol red 7.2-8.8 Yellow Red See above. a-Naphtholphthalein Cresol purple Thymol blue Phenolphthalein Thymolphthalein Alizarin yellow 7.3–8.7 Pink Green 0.1 g in 50 mL ethanol. Then add 50 mL H2O. 7.6-9.2 Yellow Purple See above. 8.0-9.6 Yellow Blue See above. 8.0-9.6 Colorless Pink 0.05 g in 50 mL ethanol. Then add 50 mL H,O. 0.04 g in 50 mL ethanol. Then add 50 mL H,O. 0.01 wt% in H,0 0.1 g in 70 mL ethanol. Then add 30 mL H,O. 0.1 wt% in H0 8.3–10.5 Colorless Blue Yellow Orange-red Orange-brown Orange 10.1–12.0 Nitramine 10.8–13.0 Colorless Tropaeolin O 11.1–12.7 Yellow
Step by Step Answer:

Related Video
A substance that alters color in solution over a constrained range of pH values is known as a pH indicator or acid-base indicator. The indicator chemical just needs to alter color slightly in order to be noticed. Indicators work on the basis that they react with water to produce the hydrogen cation H+ or hydronium ion H3O+. The indicator molecule\\\'s color changes as a result of the reaction. Since indicators have distinct color ranges for color change, they can occasionally be combined to provide color changes over a wider pH range.
Students also viewed these Engineering questions
-
Do you expect to observe more or less voluntary disclosure by companies in emerging-market countries than in developed countries? Why? Do you expect to observe more or less regulatory disclosure...
-
What type of compensation policy would you expect to observe in an industry with rapidly changing technology? Explain
-
Do you expect to see larger solidification shrinkage for a material with a bcc crystal structure or fee? Explain.
-
Panamint Candy Company prepared the following amortization table for $300,000 of 5-year, 9% bonds issued and sold by Panamint on January 1, 2021, for $285,000: Required: 1. Prepare the entry to...
-
Is it accurate to say that there is only one correct answer to every tax research question? Discuss the reasoning for your response. In what ways does your answer affect how the tax research process...
-
Write an HDL module for the Gray code counter from Exercise 3.27.? Data from problem 3.27 Gray codes have a useful property in that consecutive numbers differ in only a single bit position. Table...
-
Your colleague is excited about your good fortune (Problem 3.1) at work, but she only got the promise of a watch or \($300\) cash. You convince her that she will be better in the long run by just...
-
Relevant costs, capital budgeting, strategic decision. (M. Porporato, adapted) Wilcox is a family-owned company that has been making microwaves for almost 20 years. The companys production line...
-
What is the income summary account for? What is another name for the income summary account? What happens to the revenue and expense accounts after the closing process? Why do we close the draw...
-
Ginger Company of Bathurst has a policy of depositing all receipts and making all payments by cheque. On receiving the bank statement, Bill Free, a new bookkeeper, is quite upset that the balance in...
-
What color do you expect to observe for cresol purple indicator (Table 10-3) at the following pH values? (a) 1.0; (b) 2.0; (c) 3.0 Table 10-3 Acid color Transition Base Indicator range (pH) color...
-
Cresol red has two transition ranges listed in Table 10-3. What color would you expect it to be at the following pH values? (a) 0; (b) 1; (c) 6; (d) 9 Table 10-3 Acid color Transition Base Indicator...
-
The Bimbo Corporation has been experiencing a decline in sales relative to its major competitors. Because Bimbo is confident about the quality of its products, it suspects that this sales loss may...
-
In 2021-22, Peter is granted a 12-year lease on a property, paying a premium of 40,000 to his landlord. He immediately grants a 4-year sub-lease to Paula, receiving a premium from her of 14,000....
-
Vivian Hildenbrand and Tom Hildenbrand began living together in Oregon in 1975 and continuously did so until Toms death in 1984. During this time, pursuant to mutual agreement, they cohabited and...
-
Brenda is a company director and she earns a regular monthly salary of 6,000. In December 2021 she received a 20,000 bonus. She is provided with a diesel-engined company car (registered in January...
-
Smith is an attorney who works at the firm of Johnson & Johnson. He represents Ralph Grant, who is seeking a divorce against his wife, Alice Grant. In their first meeting, Smith learns that Ralph is...
-
Dorothy's income tax and Class 4 NICs liability for 2020-21 was 24,600, of which 600 was deducted at source. Her liability for 2021-22 is 28,000, of which none is deducted at source. She made a first...
-
On July 1, Quill & Quire magazine collected cash of $5,000 for annual subscriptions (12 issues per year) starting on August 1. Journalize the transaction to record the collection of cash on July 1...
-
Select the correct answer for each of the following questions. 1. On December 31, 20X3, Saxe Corporation was merged into Poe Corporation. In the business combination, Poe issued 200,000 shares of its...
-
(a) A luxury sports car has a frontal area of 22.4 ft 2 and a 0.29 coefficient of drag at 60 mph. What is the drag force on the vehicle at this speed? (b) A sport-utility vehicle has C D = 0.45 at 60...
-
A certain parachute has a drag coefficient of C D = 1.5. If the parachute and skydiver together weigh 225 lb, what should the frontal area of the parachute be so that the skydivers terminal velocity...
-
Submarines dive by opening vents that allow air to escape from ballast tanks and water to flow in and fill them. In addition, diving planes located at the bow are angled downward to help push the...
-
Shawn Inc. proposed to install one Robotic Machine for his new project, an initial investment of CANVAS Technology will be $520,000 and the Robotic will be expected to generate net cash flows of at...
-
You anticipate the receipt of money in 200 days, which you will use to purchase stocks in a particular company. The stock is currently selling for $51 and will pay a $0.5 dividend in 50 days and...
-
1) Based on the stock chart for Michaels Companies Inc, what do you think the short and long-term growth potentials are for this company? (discuss the advantages/disadvantages) Link to the stock...

Study smarter with the SolutionInn App



