1. Based on the information I obtained with the financial statements and 10k what would I see...
Fantastic news! We've Found the answer you've been seeking!
Question:
1. Based on the information I obtained with the financial statements and 10k what would I see as the biggest risk factor in a subsequent audit of this company?
2. What are 3 areas of audit risk that I would address in my audit?
3. What are the most surprising results I discovered about Johnson and Johnson Company?
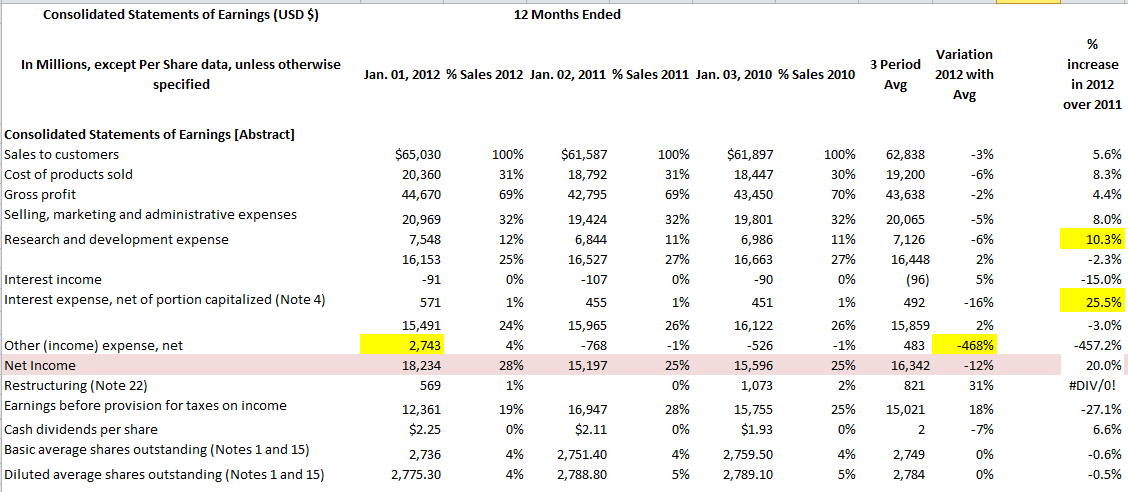
Transcribed Image Text:
Consolidated Statements of Earnings (USD $) In Millions, except Per Share data, unless otherwise specified Consolidated Statements of Earnings [Abstract] Sales to customers Cost of products sold Gross profit Selling, marketing and administrative expenses Research and development expense Interest income Interest expense, net of portion capitalized (Note 4) Other (income) expense, net Net Income Restructuring (Note 22) Earnings before provision for taxes on income Cash dividends per share Basic average shares outstanding (Notes 1 and 15) Diluted average shares outstanding (Notes 1 and 15) Jan. 01, 2012 % Sales 2012 Jan. 02, 2011 % Sales 2011 Jan. 03, 2010 % Sales 2010 $65,030 20,360 44,670 20,969 7,548 16,153 -91 571 15,491 2,743 18,234 569 12,361 $2.25 12 Months Ended 2,736 2,775.30 100% 31% 69% 32% 12% 25% 0% 1% 24% 4% 28% 1% 19% 0% 4% 4% $61,587 18,792 42,795 19,424 6,844 16,527 -107 455 15,965 -768 15,197 16,947 $2.11 2,751.40 2,788.80 100% 31% 69% 32% 11% 27% 0% 1% 26% -1% 25% 0% 28% 0% $61,897 18,447 43,450 19,801 6,986 16,663 -90 451 16,122 -526 15,596 1,073 15,755 $1.93 4% 2,759.50 5% 2,789.10 62,838 100% 30% 19,200 70% 43,638 32% 20,065 11% 7,126 27% 16,448 0% (96) 492 15,859 483 16,342 821 15,021 2 1% 26% -1% 25% 2% 25% 0% 3 Period Avg 4% 5% 2,749 2,784 Variation 2012 with Avg -3% -6% -2% -5% -6% 2% 5% -16% 2% -468% -12% 31% 18% -7% 0% 0% % increase in 2012 over 2011 5.6% 8.3% 4.4% 8.0% 10.3% -2.3% -15.0% 25.5% -3.0% -457.2% 20.0% #DIV/0! -27.1% 6.6% -0.6% -0.5% Consolidated Statements of Earnings (USD $) In Millions, except Per Share data, unless otherwise specified Consolidated Statements of Earnings [Abstract] Sales to customers Cost of products sold Gross profit Selling, marketing and administrative expenses Research and development expense Interest income Interest expense, net of portion capitalized (Note 4) Other (income) expense, net Net Income Restructuring (Note 22) Earnings before provision for taxes on income Cash dividends per share Basic average shares outstanding (Notes 1 and 15) Diluted average shares outstanding (Notes 1 and 15) Jan. 01, 2012 % Sales 2012 Jan. 02, 2011 % Sales 2011 Jan. 03, 2010 % Sales 2010 $65,030 20,360 44,670 20,969 7,548 16,153 -91 571 15,491 2,743 18,234 569 12,361 $2.25 12 Months Ended 2,736 2,775.30 100% 31% 69% 32% 12% 25% 0% 1% 24% 4% 28% 1% 19% 0% 4% 4% $61,587 18,792 42,795 19,424 6,844 16,527 -107 455 15,965 -768 15,197 16,947 $2.11 2,751.40 2,788.80 100% 31% 69% 32% 11% 27% 0% 1% 26% -1% 25% 0% 28% 0% $61,897 18,447 43,450 19,801 6,986 16,663 -90 451 16,122 -526 15,596 1,073 15,755 $1.93 4% 2,759.50 5% 2,789.10 62,838 100% 30% 19,200 70% 43,638 32% 20,065 11% 7,126 27% 16,448 0% (96) 492 15,859 483 16,342 821 15,021 2 1% 26% -1% 25% 2% 25% 0% 3 Period Avg 4% 5% 2,749 2,784 Variation 2012 with Avg -3% -6% -2% -5% -6% 2% 5% -16% 2% -468% -12% 31% 18% -7% 0% 0% % increase in 2012 over 2011 5.6% 8.3% 4.4% 8.0% 10.3% -2.3% -15.0% 25.5% -3.0% -457.2% 20.0% #DIV/0! -27.1% 6.6% -0.6% -0.5%
Expert Answer:
Answer rating: 100% (QA)
In Millions except Per Share data unless otherwise specified Jan 01 2012 Sales 2012 Jan 02 2011 Sale... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Based on the information in DDB Table 7B, identify which ideal self-concept characteristics are most versus least associated with following. Explain. a) Enjoy shopping for items influenced by other...
-
Based on the information set out in problem PA-46 below, prepare the cash flows from operating activities section of the statement of cash flows using the direct method. Data from PA-46 Tymen...
-
Based on the information provided, determine Vogls net exposure to each foreign currency in dollars.
-
Mexico discovers huge reserves of oil and starts exporting oil to the United States. Describe how this would affect the following. a. The nominal pesoU.S. dollar exchange rate. b. Mexican exports to...
-
From the nature of the carbohydrate bound to red blood cells, answer the following questions: a. People with type O blood can donate blood to anyone, but they cannot receive blood from everyone. From...
-
Condensed financial data of Sinjh SA follow. Additional information: 1. New equipment costing 80,000 was purchased for cash during the year. 2. Old equipment having an original cost of 57,500 and...
-
What are the various sources of finances for a project and which ones does your company most frequently use? Why?
-
The owners of Sweet-Tooth Bakery have determined that they need to expand their facility in order to meet their increased demand for baked goods. The decision is whether to expand now with a large...
-
Describe business ethics issues in precapitalist America. Describe ethical foundation of capitalism (Adam Smith). Discuss the balance between economic growth and stakeholder rights throughout U.S....
-
ABC Framing has been hired to frame a light commercial building. The project began on July 2 and was completed on August 9. The following is a list of accounting transactions associated with the...
-
What are the three most critical personal characteristics to look for in hiring hourly staff for a catering company? What are the three most critical personal characteristics to look for in hiring...
-
The company manufactures a single product, cleverly named Product X. The following information is available for the calendar year 2018 just completed, during which they produced and sold 200,000...
-
The Playfair cipher with keyword SWITZERLAND and matrix starting at the top center given below 71 7 71 71 71 . Find the message. 71 . K 71 71 K 71 71 were used to create the following ciphertext: ....
-
1. Simplify (3) + (6) + (2) giving your answer as a single binomial coefficient of the form 2. Find the expansion of (i) (3r-4y)4 (x - 1)8 3. Expand (a + b)5. If a = 3 and b = 1, find the value (as a...
-
PC1 is able to communicate with its default gateway, but is unable to communicate with PC2 or PC3. Determine what is preventing communication between these PCs. Since this network has been...
-
Build out a highly available website according to AWS best practices and security principals. The website servs 3 different webpages based on the path specified in the URL. These webpages are served...
-
Determine whether each relation is a function. Explain why or why not. 1. {(3,2), (-1, 3), (5, 2)} 2. {(9,-1), (3, 6), (-2, 4), (3,0)} Determine if each equation defines y as a function of x. Explain...
-
In Problems, solve each system of equations. x + 2y + 3z = 5 y + 11z = 21 5y + 9z = 13
-
write a function to calculate average_of_max_scores which takes 2D list where each row has 2 entries: a student username and a score. Allowed to make multiple submissions, so there can be multiple...
-
a. What are some of the major objectives of the IMF? b. How is the IMF involved in international trade?
-
Fairfield Corp., a U.S. firm, recently established a subsidiary in a less developed country that consistently experiences an annual inflation rate of 80 percent or more. The country does not have an...
-
Orlando Co. has its U.S. business funded with dollars with a capital structure of 60 percent debt and 40 percent equity. It has its Thailand business funded with Thai baht with a capital structure of...
-
The heat of vaporization of ether is \(25.98 \mathrm{~kJ} / \mathrm{mol}\) at its boiling point, \(34.5^{\circ} \mathrm{C}\). (a) Calculate the rate of change of vapour pressure with temperature...
-
For a gas obeying the equation of state \(V=B+\frac{R T}{P}\), the Joule-Thomson coefficient is given by \[ \mu_{\mathrm{JT}}=-\frac{1}{C_{P}}\left(T \frac{d B}{d T}-B ight) \]
-
Calculate the residual enthalpy and entropy for carbon dioxide at \(393 \mathrm{~K}\) and \(12 \mathrm{MPa}\) using any equation of state.

Study smarter with the SolutionInn App


