Question: Consider the biosensor device shown in the figure below. The biosensor is designed to measure the concentration of solute A in the well-mixed liquid phase.
Consider the biosensor device shown in the figure below. The biosensor is designed to measure the concentration of solute A in the well-mixed liquid phase. At the base of the decice is an electrode of surface area 2.0 cm2. The electrode is coated with an enzyme that catalyzes the reaction A ? 2D. When solute A reacts to product D, product D is detected by the electrode, enabling for direct measurement of the flux of the product D, which at steady state can be used to determine the concentration of A in the bulk liquid. The rate of reaction of A at the enzyme surface is rapid relative to the rate of diffusion of A down to the surface. Directly above the enzyme-coated electrode is a gel layer of 0.30 cm thickness that serves as a diffusion barrier for solute A and protects the enzyme. The gel layer is designed to make the flux of A down to the enzyme-coated surface diffusion limited. The effective diffusion coefficient of solute A in this gel layer is DAe = 4.0 ? 10-7 cm2/s at 20?C. Above the gel layer is a well mixed liquid containing a constant concentration of solute A, C?Ao. The solubility of solute A in the liquid differs from the solubility of A in the gel layer. Specifically, the equilibrium partitioning constant K = 0.8 cm3 gel / cm3 liquid. The process is considered very dilute, and the total molar concentration of the gel layer is unknown. The concentration of product D in the wellmixed liquid is very small so that cDo ? 0. At 20?C, the electrode measures that the formation of product D is equal to 3.6 ? 10?5 mmole D/h. What is the concentration of solute A in the bulk well-mixed liquid phase, C?Ao, in units of mmole/cm3?
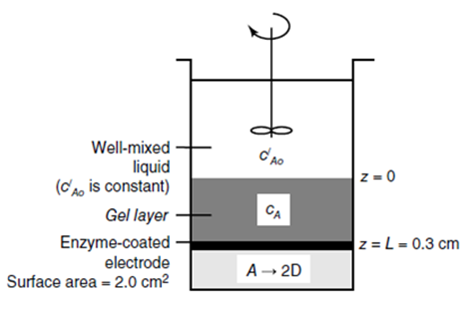
Well-mixed liquid (CAD is constant) Gel layer Enzyme-coated electrode Surface area = 2.0 cm f C AO CA A 2D z=0 z=L=0.3 cm
Step by Step Solution
3.42 Rating (152 Votes )
There are 3 Steps involved in it
To find the concentration of solute A in the bulk wellmixed liquid phase CAo we can use Ficks first ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (2 attachments)
6091cc20c4ea2_22594.pdf
180 KBs PDF File
6091cc20c4ea2_22594.docx
120 KBs Word File


