1. (40 pts) Citric acid (C6H8O7) is used in the preparation of many foods, pharmaceuticals, and...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
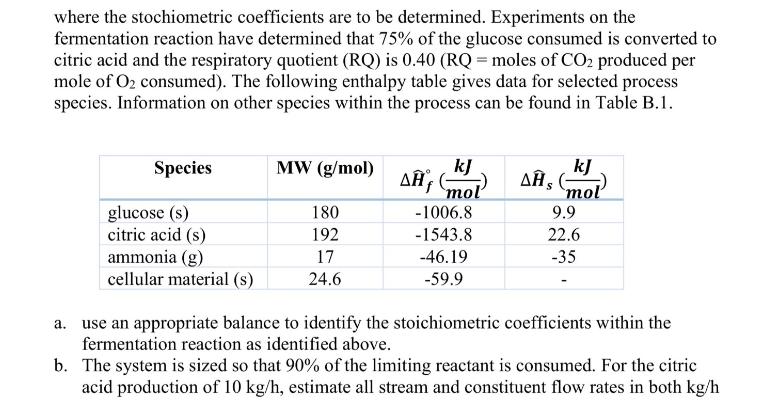
1. (40 pts) Citric acid (C6H8O7) is used in the preparation of many foods, pharmaceuticals, and personal care products. Although it can be recovered by concentration and crystallization from citrus juices, modern commercial production involves synthesis by fermentation of carbohydrates by Aspergillus niger. The process involves addition of the fungus to a fermenter along with carbohydrates, nutrients, water, and air that is bubbled through the fermentation broth. After the desired production of citric acid, the resultant fermentation liquor is filtered to remove the cell mass and other suspended solids followed by recovery and purification of citric acid by crystallization. As part of the evaluation of a proposed continuous fermentation process, you have been tasked with estimation of the required heating/cooling requirements for the fermenter designed to produce 10 kg/h of citric acid. Feed into the unit includes an aqueous solution that is 20 wt.% glucose, 0.4wt% ammonia, and the balance water. Air at 1.2 atm, saturated with water, is bubbled through fermenter providing a molar flowrate of oxygen twice that of glucose. One major stream leaving the fermenter is a gas stream at 1 atm containing nitrogen, the unreacted oxygen, carbon dioxide produced during the fermentation process. This gas stream is also saturated with water. The other major stream leaving the fermenter is a liquid fermentation liquor containing water, citric acid, unreacted glucose, and unreacted ammonia. All streams are assumed to exist at 25 C and the contents of the fermenter can be assumed to be well mixed due to the nature of air bubbling within the fermenter. The fermentation reaction is given as: C6H12O6 (aq) + a NH3(aq) + b O(9) C CH1.79N0.200.5 (s) + d CO2 + e HO (1) + f C6H8O7 (aq) where the stochiometric coefficients are to be determined. Experiments on the fermentation reaction have determined that 75% of the glucose consumed is converted to citric acid and the respiratory quotient (RQ) is 0.40 (RQ = moles of CO2 produced per mole of O2 consumed). The following enthalpy table gives data for selected process species. Information on other species within the process can be found in Table B.1. Species MW (g/mol) kJ kJ A mol , (mol glucose (s) citric acid (s) 180 -1006.8 9.9 192 -1543.8 22.6 ammonia (g) 17 -46.19 -35 cellular material (s) 24.6 -59.9 a. use an appropriate balance to identify the stoichiometric coefficients within the fermentation reaction as identified above. b. The system is sized so that 90% of the limiting reactant is consumed. For the citric acid production of 10 kg/h, estimate all stream and constituent flow rates in both kg/h and mol/h. What are the volumetric flow rates of air entering the fermenter and the off-gas stream (in L/h)? c. The heats of formation provided within the table indicate the phases of materials; however, the reaction is specified with substances within the aqueous phase. Show how Hess's law can be used to estimate the heat of formation in the aqueous phase based on the provided enthalpies for the gaseous and solid species. d. Estimate the rate at which heat must be added or removed from the fermenter (in kW). 1. (40 pts) Citric acid (C6H8O7) is used in the preparation of many foods, pharmaceuticals, and personal care products. Although it can be recovered by concentration and crystallization from citrus juices, modern commercial production involves synthesis by fermentation of carbohydrates by Aspergillus niger. The process involves addition of the fungus to a fermenter along with carbohydrates, nutrients, water, and air that is bubbled through the fermentation broth. After the desired production of citric acid, the resultant fermentation liquor is filtered to remove the cell mass and other suspended solids followed by recovery and purification of citric acid by crystallization. As part of the evaluation of a proposed continuous fermentation process, you have been tasked with estimation of the required heating/cooling requirements for the fermenter designed to produce 10 kg/h of citric acid. Feed into the unit includes an aqueous solution that is 20 wt.% glucose, 0.4wt% ammonia, and the balance water. Air at 1.2 atm, saturated with water, is bubbled through fermenter providing a molar flowrate of oxygen twice that of glucose. One major stream leaving the fermenter is a gas stream at 1 atm containing nitrogen, the unreacted oxygen, carbon dioxide produced during the fermentation process. This gas stream is also saturated with water. The other major stream leaving the fermenter is a liquid fermentation liquor containing water, citric acid, unreacted glucose, and unreacted ammonia. All streams are assumed to exist at 25 C and the contents of the fermenter can be assumed to be well mixed due to the nature of air bubbling within the fermenter. The fermentation reaction is given as: C6H12O6 (aq) + a NH3(aq) + b O(9) C CH1.79N0.200.5 (s) + d CO2 + e HO (1) + f C6H8O7 (aq) where the stochiometric coefficients are to be determined. Experiments on the fermentation reaction have determined that 75% of the glucose consumed is converted to citric acid and the respiratory quotient (RQ) is 0.40 (RQ = moles of CO2 produced per mole of O2 consumed). The following enthalpy table gives data for selected process species. Information on other species within the process can be found in Table B.1. Species MW (g/mol) kJ kJ A mol , (mol glucose (s) citric acid (s) 180 -1006.8 9.9 192 -1543.8 22.6 ammonia (g) 17 -46.19 -35 cellular material (s) 24.6 -59.9 a. use an appropriate balance to identify the stoichiometric coefficients within the fermentation reaction as identified above. b. The system is sized so that 90% of the limiting reactant is consumed. For the citric acid production of 10 kg/h, estimate all stream and constituent flow rates in both kg/h and mol/h. What are the volumetric flow rates of air entering the fermenter and the off-gas stream (in L/h)? c. The heats of formation provided within the table indicate the phases of materials; however, the reaction is specified with substances within the aqueous phase. Show how Hess's law can be used to estimate the heat of formation in the aqueous phase based on the provided enthalpies for the gaseous and solid species. d. Estimate the rate at which heat must be added or removed from the fermenter (in kW).
Expert Answer:

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, ? Ronald W. Rousseau, ? Lisa G. Bullard
Posted Date:
Students also viewed these chemical engineering questions
-
Citric acid (C 6 H 8 O 7 ) is used in the preparation of many foods, pharmaceuticals, soft drinks, and personal-care products. Although it can be recovered by concentration and crystallization from...
-
"internet radios" for streaming audio, and personal video recorders and players. Describe design and evaluation processes that could be used by a start-up company to improve the usability of such...
-
Uniform rod AB of length l and mass m lies in a vertical plane and is acted upon by a couple M. The ends of the rod are connected to small rollers which rest against frictionless surfaces. (a)...
-
Kilgore Company makes and sells a single product. Kilgore incurred the following costs in its most recent fiscal year: Cost Items Appearing on the Income Statement Materials cost ($7 per...
-
Describe how organizations can manage work-family conflicts?
-
A construction contract has the following language: It is the responsibility of the contractor to inspect and become familiar with the Project and to acquaint itself thoroughly with all conditions...
-
Internal Indexes?Dollar-Value LIFO On January 1, 2010, Bonanza Wholesalers Inc. adopted the dollar-value LIFO inventory method for income tax and external financial reporting purposes. However,...
-
Explain the difference between " management controls ", which are generally the responsibility of the client's Management team and " transaction controls ", which are typically performed by...
-
APPENDIX D Problem 7-06 A widow currently has a $84,000 investment that yields 6 percent annually. Can she withdraw $13,000 for the next ten years? Use Appendix D to answer the question. Round your...
-
A service company is likely to benefit from using which types of variances? A. Labor variances but not material and overhead variances B. Labor and overhead variances but not material variances
-
Which of these is How can an s-curve be misinterpreted? It can't be misinterpreted An increase in costs due to expensive material being accrued on the project before they should Follow the s-curve to...
-
You are serving fresh figs with goat cheese. Each serving will contain fresh fig and 1 tablespoon of goat cheese. Figs are purchased by the case, 35 figs per case, for $25.90. The yield percent for...
-
What do we mean when we talk about standardization, convergence or harmonization of accounting standards? Do they mean m,ism? From the point of view of a U.S. company with operations abroad, what are...
-
A home located at 1323 Spruce Street has been sold for $400,000. The buyer has given the broker $5,000 as earnest money. The seller will pay the 6% real estate brokerage commission, the buyer's title...
-
Find the short circuit current The circuit you should use to find the short circuit current, i sc, is shown here. Note that the load resistor to the right of the terminals a-b has been replaced by a...
-
Arlington Merchants reported the following on its income statement for the fiscal years ending December 31, 2016 and 2015. 2016 2015 Sales $4,857,500 $4,752,900 Cost of goods sold 3,258,950 3,207,000...
-
Rain is falling on a poorly designed 150-m2 flat roof of a contemporary home. The design flaw requires the water level on the roof to reach approximately 5 cm above the plane of the roof before the...
-
Several decades ago benzene was thought to be a harmless chemical with a somewhat pleasant odor and was widely used as a cleaning solvent. It has since been found that chronic exposure to benzene can...
-
As described in Problem 9.21, the manufacture of ethanol from corn starch involves fermentation using a yeast that converts sugars from the starch to ethanol and carbon dioxide in a complicated...
-
At a point \(P\) in a body, Cartesian stress components are given by \(\sigma_{x x}=80 \mathrm{MPa}, \sigma_{y y}=-40\) \(\mathrm{MPa}, \sigma_{z z}=-40 \mathrm{MPa}\), and \(\tau_{x y}=\tau_{y...
-
Find the principal stresses and the orientation of the principal axes of stresses for the following cases of plane stress. a. \(\sigma_{x x}=40 \mathrm{MPa}, \sigma_{y y}=0 \mathrm{MPa}, \tau_{x...
-
Determine the principal stresses and their associated directions, when the stress matrix at a point is given by 11 1 [6]=112 MPa. [121]

Study smarter with the SolutionInn App


