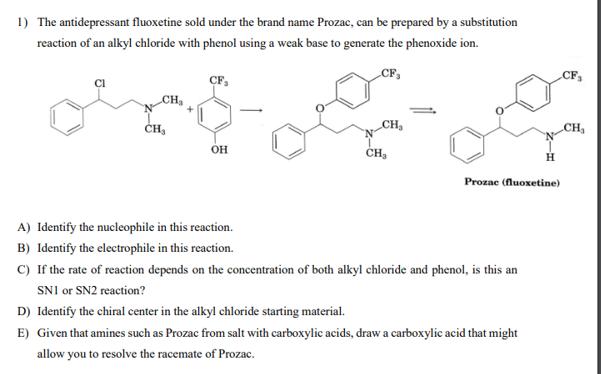
1) The antidepressant fluoxetine sold under the brand name Prozac, can be prepared by a substitution...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
1) The antidepressant fluoxetine sold under the brand name Prozac, can be prepared by a substitution reaction of an alkyl chloride with phenol using a weak base to generate the phenoxide ion. CF, CH₂ CF₂ OH A) Identify the nucleophile in this reaction. B) Identify the electrophile in this reaction. CH₂ CH₂ H Prozac (fluoxetine) C) If the rate of reaction depends on the concentration of both alkyl chloride and phenol, is this an SNI or SN2 reaction? D) Identify the chiral center in the alkyl chloride starting material. E) Given that amines such as Prozac from salt with carboxylic acids, draw a carboxylic acid that might allow you to resolve the racemate of Prozac. CF₁ CH₁ 1) The antidepressant fluoxetine sold under the brand name Prozac, can be prepared by a substitution reaction of an alkyl chloride with phenol using a weak base to generate the phenoxide ion. CF, CH₂ CF₂ OH A) Identify the nucleophile in this reaction. B) Identify the electrophile in this reaction. CH₂ CH₂ H Prozac (fluoxetine) C) If the rate of reaction depends on the concentration of both alkyl chloride and phenol, is this an SNI or SN2 reaction? D) Identify the chiral center in the alkyl chloride starting material. E) Given that amines such as Prozac from salt with carboxylic acids, draw a carboxylic acid that might allow you to resolve the racemate of Prozac. CF₁ CH₁
Expert Answer:
Answer rating: 100% (QA)
A The nucleophile in this reaction is OH as O is the most electr... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Fluoxetine, a heavily prescribed antidepressant marketed under the name Prozac, can be prepared by a route that begins with reaction between a phenol and an alkyl chloride. (a) The rate of the...
-
The reaction of an alkyl chloride with potassium iodide is generally carried out in acetone to maximize the amount of alkyl iodide that is formed. Why does the solvent increase the yield of alkyl...
-
From the following facts, calculate the correct cost of inventory for Sue Company. Cost of inventory on shelf, $4,500, which includes $280 of goods received on consignment. Goods in transit en...
-
An article in Barron's noted the following. Okay. Last fall, someone with a long memory and an even longer arm reached into that bureau drawer and came out with a moldy cheese sandwich and the...
-
SFAC No. 5 states that the concept of capital maintenance is critical in distinguishing an enterprises return on investment from return of its investment. Two concepts of capital maintenance are...
-
Discuss the privacy and confidentiality issues of HIV-positive patients.
-
Black Rose Company has always done some planning for the future, but the company has never prepared a formal budget. Now that the company is growing larger, it is considering preparing a budget....
-
Laval produces lighting fixtures. Budgeted information for its two production departments follows. The departments use machine hours (MH) and direct labor hours (DLH). Overhead cost Fabricating $...
-
Imagine that you are Magna's new corporate controller and answer the following: 1. Describe Magna's strategy in terms of how it competes for customers. 2. Based on Magna's strategy and the data...
-
What is the change in internal energy (in J) of a system that releases 675 J of thermal energy to its surroundings and has 9.60 x 10 cal of work done on it? Give your answer in scientific notation. ...
-
What are the reasons for limitation periods generally? How do they apply to land?
-
Distinguish between the circumstances under which an easement may be obtained by prescription and a title may be extinguished by adverse possession.
-
What normally constitutes the consideration for a guarantee?
-
Describe the two main classifications of interests in land. Distinguish between freehold and leasehold estates.
-
To whom does a guarantor give his or her promise?
-
Jason Corporation has invested in a machine that cost $78,000, that has a useful life of six years, and that has no salvage value at the end of its useful life. The machine is being depreciated by...
-
In muscle tissue, the ratio of phosphorylase a to phosphorylase b determines the rate of conversion of glycogen to glucose 1phosphate. Classify how each event affects the rate of glycogen breakdown...
-
Propose a structure for an aromatic hydrocarbon, C 11 H 16 that has the following 13 C NMR spectral data: Broadband-decoupled 13 C NMR: 29.5, 31.8, 50.2, 125.5, 127.5, 130.3, 139.8 DEPT-90: 125.5,...
-
Proteins can be cleaved specifically at the amide bond on the carboxyl side of methionine residues by reaction with cyanogen bromide, BrC = N. The reaction occurs in several steps: (a) The first step...
-
Aspartame, a nonnutritive sweetener marketed under the trade name NutraSweet (among others), is the methyl ester of a simple dipeptide, Asp-Phe-OCH3. (a) Draw the structure of aspartame. (b) The...
-
If the 146 customer visits is used to estimate the number of appliance department visits at the larger store population, what are the lower and upper limits of this estimate?
-
What proportion of the 26,280 individuals are in each of the five categories?
-
What are the percentages of observations in each of the five job satisfaction categories?

Study smarter with the SolutionInn App


