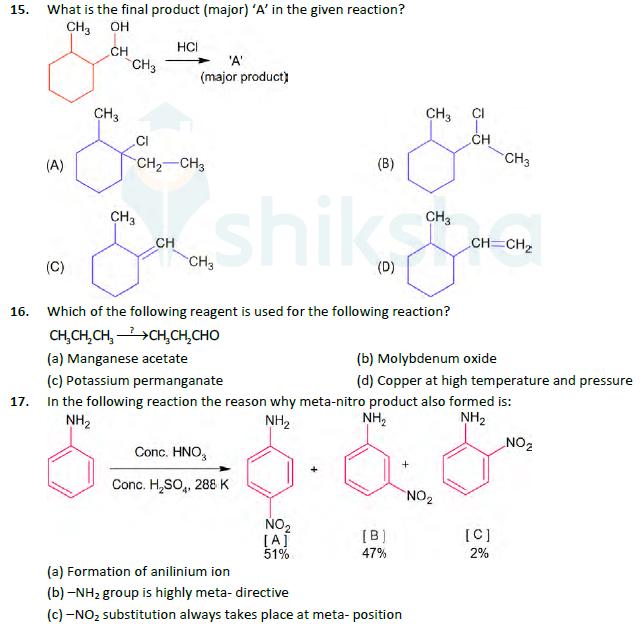
15. 16. What is the final product (major) 'A' in the given reaction? CH3 OH CH...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
15. 16. What is the final product (major) 'A' in the given reaction? CH3 OH CH CH3 HCI + 'A' (major product) CH3 CH3 Cl CH (B) CH3 CH2-CH3 (A) CH3 CH (C) shiks CH=CH2 (D) Which of the following reagent is used for the following reaction? CHCH,CH, +CHCH,CHO (a) Manganese acetate (b) Molybdenum oxide (c) Potassium permanganate (d) Copper at high temperature and pressure 17. In the following reaction the reason why meta-nitro product also formed is: NH2 NH2 NH2 NH2 NO2 Conc. HNO3 + Conc. HSO, 288 K NO2 NO2 [A] [B] [C] 51% 47% 2% (a) Formation of anilinium ion (b)-NH2 group is highly meta- directive (c) -NO substitution always takes place at meta- position 18. (d) low temperature Which of the following are isostructural pairs? A. SO and Cro B. SiCl4 and TiCl4 C. NH3 and NO D. BCl3 and BrCl 3 (a) C and D only (b) A and B only (c) A and C only (d) B and C only 19. Out of the following which type of interaction is responsible for the stabilization of -helix structure of proteins? (a) Hydrogen bonding (b) Covalent bonding (c) Vander Waals forces (d) lonic bonding 20. The gas released during anaerobic degradation of vegetation may lead to: (a) Ozone hole (c) Corrosion of metals (b) Global warming and cancer (b) Acid rain 15. 16. What is the final product (major) 'A' in the given reaction? CH3 OH CH CH3 HCI + 'A' (major product) CH3 CH3 Cl CH (B) CH3 CH2-CH3 (A) CH3 CH (C) shiks CH=CH2 (D) Which of the following reagent is used for the following reaction? CHCH,CH, +CHCH,CHO (a) Manganese acetate (b) Molybdenum oxide (c) Potassium permanganate (d) Copper at high temperature and pressure 17. In the following reaction the reason why meta-nitro product also formed is: NH2 NH2 NH2 NH2 NO2 Conc. HNO3 + Conc. HSO, 288 K NO2 NO2 [A] [B] [C] 51% 47% 2% (a) Formation of anilinium ion (b)-NH2 group is highly meta- directive (c) -NO substitution always takes place at meta- position 18. (d) low temperature Which of the following are isostructural pairs? A. SO and Cro B. SiCl4 and TiCl4 C. NH3 and NO D. BCl3 and BrCl 3 (a) C and D only (b) A and B only (c) A and C only (d) B and C only 19. Out of the following which type of interaction is responsible for the stabilization of -helix structure of proteins? (a) Hydrogen bonding (b) Covalent bonding (c) Vander Waals forces (d) lonic bonding 20. The gas released during anaerobic degradation of vegetation may lead to: (a) Ozone hole (c) Corrosion of metals (b) Global warming and cancer (b) Acid rain
Expert Answer:
Answer rating: 100% (QA)
Q15 Correct Option A The first step in the mechanism involves protonation of the alcoholic Oatom followed by the removal of water leading to a seconda... View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Posted Date:
Students also viewed these chemical engineering questions
-
What does it mean with joint tenancy (where tenants own same interest) and tenants in common (where tenants own distinct interests) that each tenant has a right of possession of the whole estate?...
-
Weatherford International The oilfield services industry includes thousands of companies large and small that provide drilling, seismic testing, transportation, and a wide range of other services to...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
What approaches to recruitment can an employer adopt in order to create and project a positive public image?
-
Formamide (HCONH2) has a pKa of approximately 25. Predict, based on the map of electrostatic potential for formamide shown here, which hydrogen atom(s) has this pKa value. Support your conclusion...
-
Internal Rate of Return Juan Gonzales, the president of Nogalis Corporation, is trying to decide whether he should buy a new machine that will improve production efficiency. The machine will increase...
-
Why closing stock is not shown in the trial balance?
-
Rockport Municipal Marina is considering adding a new dock to accommodate large yachts. The dock would cost $700,000 and would generate $144,000 annually in new cash inflows. Its expected life would...
-
A) B) B) (CONTINUED) C AND D) I've already done the journal entries but i need b, c and d thanks The Di Vidnd Corporation was incorporated on January 2, 2020, with two classes of share capital: an...
-
A contractor wants to learn more about mitigation supply chain risks for federal information systems. What publication should they refer to
-
Q3. (a) Sketch and name the road structure layers including the bituminous coating layers. State the material's CBR value and the degree of compaction (DOC) for each road layer required by road works...
-
2. Justify the following in terms of impulse and momentum: a. Why are padded dashboards safer in automobiles?
-
The kidneys are an essential organ to regulating blood pressure in the cardiovascular system. Renal epithelial cells feature cilia on their surface, which allows them to sense blood flow in the...
-
Novak Corp. supplies its customers with high-quality canvas tents. These canvas tents sell for $170 each, with the following DM and DL usage and price expectations. Direct materials 9 square yards...
-
Develop a one-week schedule for your Housekeeping employees. Using the Excel spreadsheet that is attached above, you will prepare a schedule for the week The day-by-day occupied rooms forecast has...
-
On July 1, 2021, Bird Inc, a private enterprise, acquired 1,250 shares of Duck Ltd. for $75,000. This investment represents a 14% interest in Duck Ltd. and Bird Inc. uses the cost method to record...
-
1. Below is depicted a graph G constructed by joining two opposite vertices of C12. Some authors call this a "theta graph" because it resembles the Greek letter 0. a. What is the total degree of this...
-
Think of a firm that successfully achieved a combination overall cost leadership and differentiation strategy. What can be learned from this example? Are the advantages sustainable? Why? Why not?...
-
Web-based exercise. Search the Web for a recent poll in which the sample statistic is a proportion, for example, the proportion in the sample responding Yes to a question. Calculate a 95% confidence...
-
A sampling distribution, continued. Exercise 21.33 presents 50 sample means x from 50 random samples of size 100. Using a calculator, find the mean and standard deviation of these 50 values. Then...

Study smarter with the SolutionInn App


