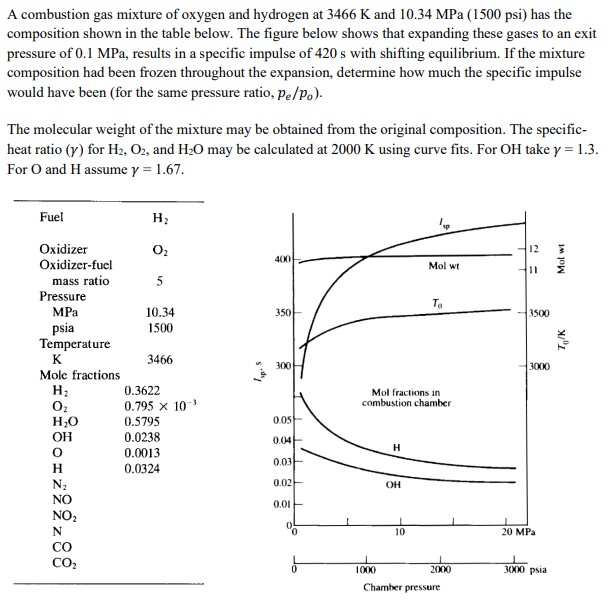
A combustion gas mixture of oxygen and hydrogen at 3466 K and 10.34 MPa (1500 psi)...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A combustion gas mixture of oxygen and hydrogen at 3466 K and 10.34 MPa (1500 psi) has the composition shown in the table below. The figure below shows that expanding these gases to an exit pressure of 0.1 MPa, results in a specific impulse of 420 s with shifting equilibrium. If the mixture composition had been frozen throughout the expansion, determine how much the specific impulse would have been (for the same pressure ratio, pe/Po). The molecular weight of the mixture may be obtained from the original composition. The specific- heat ratio (y) for H2, O2, and H2O may be calculated at 2000 K using curve fits. For OH take y = 1.3. For O and H assume y = 1.67. Mol wt Fuel Oxidizer H 02 12 400 Oxidizer-fuel Mol wt 11 mass ratio 5 Pressure To MPa 10.34 350 3500 psia 1500 Temperature K 3466 300 3000 Mole fractions H 0.3622 Oz 0.795 x 10-3 Mol fractions in combustion chamber HO 0.5795 0.05 OH 0.0238 0.04- H 0.0013 0.03 H 0.0324 N 0.02 OH NO 0.01 NO2 N CO CO 1000 10 Chamber pressure 20 MPa 2000 3000 psia To/K A combustion gas mixture of oxygen and hydrogen at 3466 K and 10.34 MPa (1500 psi) has the composition shown in the table below. The figure below shows that expanding these gases to an exit pressure of 0.1 MPa, results in a specific impulse of 420 s with shifting equilibrium. If the mixture composition had been frozen throughout the expansion, determine how much the specific impulse would have been (for the same pressure ratio, pe/Po). The molecular weight of the mixture may be obtained from the original composition. The specific- heat ratio (y) for H2, O2, and H2O may be calculated at 2000 K using curve fits. For OH take y = 1.3. For O and H assume y = 1.67. Mol wt Fuel Oxidizer H 02 12 400 Oxidizer-fuel Mol wt 11 mass ratio 5 Pressure To MPa 10.34 350 3500 psia 1500 Temperature K 3466 300 3000 Mole fractions H 0.3622 Oz 0.795 x 10-3 Mol fractions in combustion chamber HO 0.5795 0.05 OH 0.0238 0.04- H 0.0013 0.03 H 0.0324 N 0.02 OH NO 0.01 NO2 N CO CO 1000 10 Chamber pressure 20 MPa 2000 3000 psia To/K
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
Coral Divers Resort Jonathon Greywell locked the door on the equipment shed and began walking back along the boat dock to his office. He was thinking about the matters that had weighed heavily on his...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Explain, in your own words, what is meant by source inspection.
-
At December 31, 2017, Ashley Co. has outstanding purchase commitments for 150,000 gallons, at $6.20 per gallon, of a raw material to be used in its manufacturing process. The company prices its raw...
-
Truefalse A truefalse test consists of 50 questions. How many does a student have to get right to convince you that he is not merely guessing? Explain.
-
1. Identify 3 students to play the roles of the employees. Ask these 3 individuals to read their roles below. 2. Identify 1 student to play the role of the president of the social enterprise (Taylor...
-
Customer profitability, distribution. Spring Distribution has decided to analyze the profitability of five new customers. It buys bottled water at $12 per case and sells to retail customers at a list...
-
5. Suppose that acts on density operators for Q with Kraus representation () - k=1 Let U M, (C), and define operators = 1, k BkUkjAj. , Show that B,..., B., are also Kraus operators for E. 6. Even...
-
Furniture Mart manufactures and sells a range of furniture. The company follows a just- in-time production philosophy, which means that it only buys materials on a need basis for production and it...
-
Warren, a division manager of Walker Enterprises, is under pressure to boost the performance of the Lighting Division in 2014. Unfortunately, recent profits have not met expectations. The expected...
-
Describe and explain the process needed to add a new physical hard drive to a system and use the new space to make and mount a new logical volume. Include as much detail as possible including any and...
-
Aztec Clay Distributor is a family owned business that is owned by Alice, Bill, Chad and Zion. Alice and Bill are over 50. Zion is not an employee, rather a silent or somewhat silent partner. Alice...
-
How are critical access hospitals (CAHs) paid by Medicare?
-
You have a $3,000 semi-annual 1.250% bond maturing in exactly 26 years. If the bond is quoted at 58.7075 then the purchase price of the bond is $ 1,761.23 and the yield to maturity is rounded three...
-
Set down a paper on Continuing Education Program for Engineers.
-
The column shown in the figure is fixed at the base and free at the upper end. A compressive load P acts at the top of the column with an eccentricity e from the axis of the column. Beginning with...
-
What is Stirlings approximation? Why is it useful? When is it applicable?
-
Explain why T 1 T 2 .
-
The following images show contours of constant electron density for H 2 calculated using the methods described in Chapter 26. The values of electron density are (a) 0.10, (b) 0.15, (c) 0.20, (d)...
-
The SDLC is just one model for systems development. Find at least one more and describe the differences.
-
Draw DFDs for each of these scenarios: (a) A customer goes into a bookshop and asks for this book. The member of staff looks for the book in the online stock catalogue and reports that the book is...
-
Draw an entity model to model this university scenario: A university department employs lecturers and clerical staff. It offers a three-year degree. A student has to take 12 modules during the...

Study smarter with the SolutionInn App


