A Co-S-H2O Eh-pH diagram involving CoS as the only cobalt sulfide species is provided below. An expanded
Question:
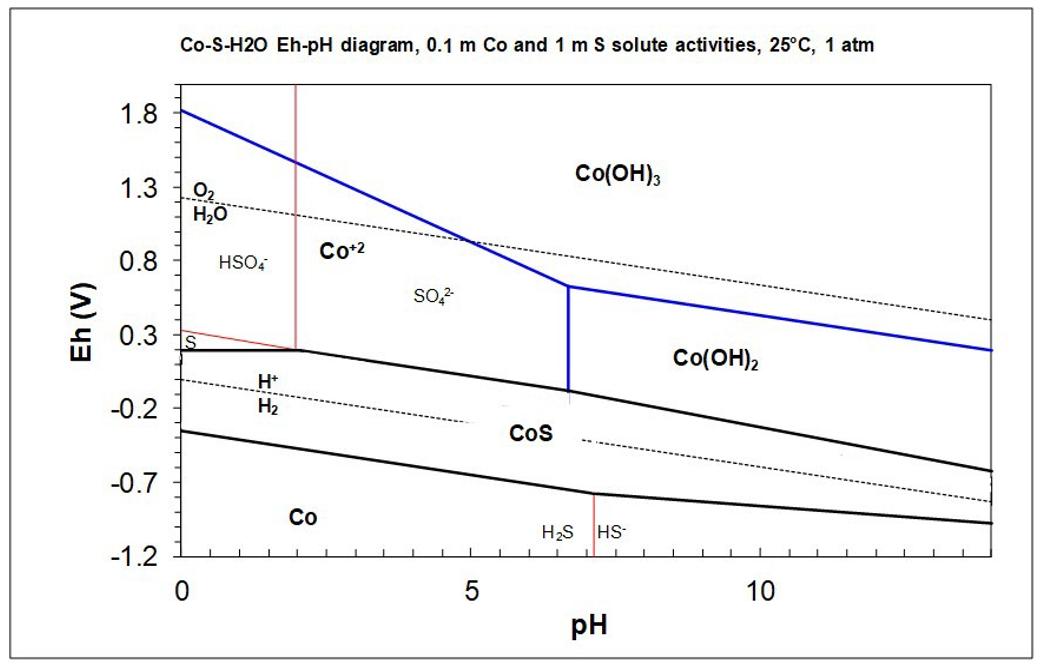
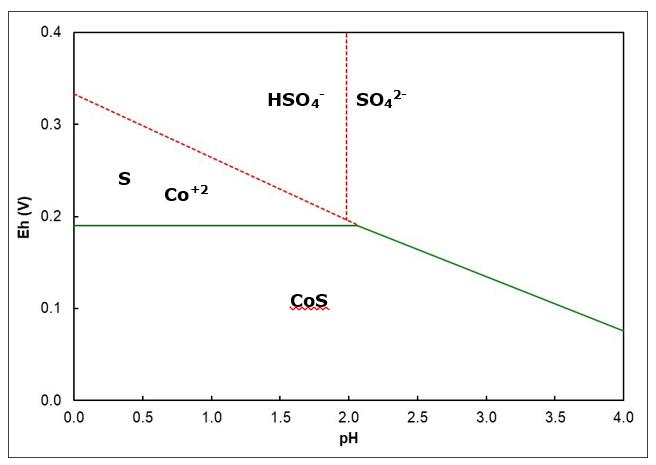
1. In principle, can a mixture of ferric and ferrous sulfates be used to leach CoS at pH 1 (Eo = 0.68 V)?
2. Indicate all possible pairs of Co and S species that can be formed by leaching CoS. Sulfur products may be soluble or insoluble.
3. Over what pH range can CoS be leached to dissolve cobalt under these conditions (as per the diagram)?
4. Estimate the concentration of ferric ion in contact with Fe(OH)3 at pH 5. The Ksp for Fe(OH)3 is 1.6 x 10-39.
5. Do you think it is feasible to use ferric sulfate to leach CoS at pH 5? Explain.
6. Equations for several Eh lines are provided in the table below in the form Eh = slope x pH + constant. Consider the leaching of CoS by oxygen gas in air (air contains 21% oxygen) at 1 atm pressure, 25oC and pH = 2.5. Calculate the Eh for the O2/H2O half reaction and for the appropriate cobalt-containing half reaction. Report to three decimal places. Include the units. Calculate ∆E for the reaction (and report the units).
7. Is the reaction favourable? Explain briefly.
Half reaction | Slope | Constant |
Co+2/Co |
| -0.3116 |
Co(OH)3/Co+2 | -0.1775 | 1.8163 |
Co+2, S/CoS |
| 0.1899 |
Co+2, SO42-/CoS | -0.05917 | 0.3121 |
Co(OH)2, SO42-/CoS | -0.07396 | 0.4110 |
HSO4-/S | -0.06903 | 0.3333 |
SO42-/S | -0.07889 | 0.3528 |

Introduction To Actuarial And Financial Mathematical Methods
ISBN: 9780128001561
1st Edition
Authors: Stephen Garrett




