A hydrometer of a given weight will sink when placed in a fluid, and the amount...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
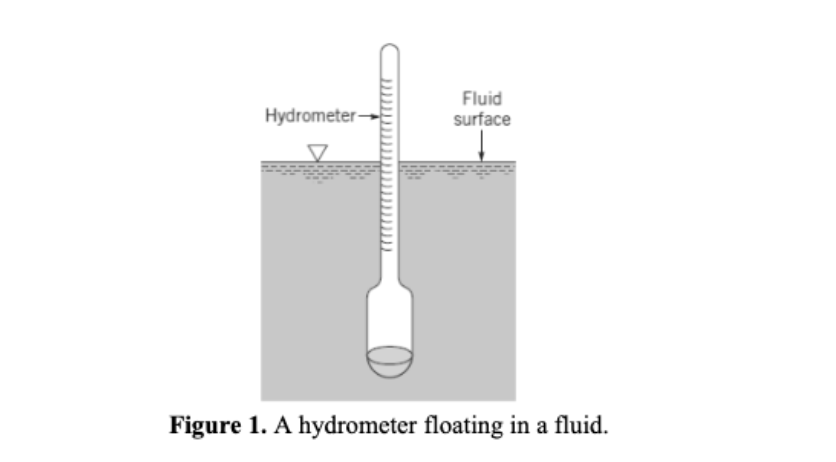
A hydrometer of a given weight will sink when placed in a fluid, and the amount it sinks is based on the fluid density. The hydrometer shown in the figure below has a mass, m, of 0.09 kg, and the cross-sectional area, Acs, of its stem is 290 mm. The hydrometer is neutrally buoyant when placed in a fluid with S.G. = 1.0. a. Will the hydrometer rise or sink when placed in a fluid of S.G. 0.9? Calculate by how much (in mm). Be sure to draw a free body diagram and a force balance. b. The highest gradation mark on the stem of the hydrometer is 15 cm above the lowest. If the smallest S.G. the hydrometer can measure is 0.7, what is greatest S.G. that this hydrometer can measure? (Hint: Is the S.G. = 0.7 reading at the top or bottom of the hydrometer?) Hydrometer- Fluid surface Figure 1. A hydrometer floating in a fluid. A hydrometer of a given weight will sink when placed in a fluid, and the amount it sinks is based on the fluid density. The hydrometer shown in the figure below has a mass, m, of 0.09 kg, and the cross-sectional area, Acs, of its stem is 290 mm. The hydrometer is neutrally buoyant when placed in a fluid with S.G. = 1.0. a. Will the hydrometer rise or sink when placed in a fluid of S.G. 0.9? Calculate by how much (in mm). Be sure to draw a free body diagram and a force balance. b. The highest gradation mark on the stem of the hydrometer is 15 cm above the lowest. If the smallest S.G. the hydrometer can measure is 0.7, what is greatest S.G. that this hydrometer can measure? (Hint: Is the S.G. = 0.7 reading at the top or bottom of the hydrometer?) Hydrometer- Fluid surface Figure 1. A hydrometer floating in a fluid.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
CANMNMM January of this year. (a) Each item will be held in a record. Describe all the data structures that must refer to these records to implement the required functionality. Describe all the...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
9. A molybdenum-vanadium alloy of composition 50wt%Mo - 50wt%V is slowly cooled from a temperature of 2600C to 1800C. Determine: a) At what temperature does the first solid phase form? b) What is the...
-
Outback Cycles, Inc., reported a number of special items on its income statement. The following data, listed in no particular order, came from Outback's financial statements (amounts in thousands):...
-
In Problem match each of the functions with one of the graphs labeled (a)(l) shown following these functions. Recognizing special features of certain types of functions and plotting points for the...
-
T. Christian Cooper was a partner to Sanders and Richard Campbell d/b/a The Mullen Company. In 2001, Cooper helped bring about a management agreement between The Mullen Co. and Newnan Crossing...
-
The following data are from the income statements of Haskin Company. (a) Compute for each year (1) The inventory turnover (2) The days in inventory. (b) What conclusions concerning the management of...
-
Discuss similarities and differences of A. MongoDB, B. Amazon (AWS) database management system to relational database management systems. Indicate the benefits or disadvantages you associate with the...
-
Ms. Ankita completed her MBA from DAVV, Indore. She joined an enterprise as an Accounts Manager at a CTC of Rs 650000. She invested Rs 150000 in PPF and paid mediclaim for herself Rs 15000 using cash...
-
Do you think companies should monitor employees' social media? Why or why not?
-
Question 2. Outline four (4) techniques to deal with customers and two (2) techniques to identify needs of customers with special needs.
-
Discuss the 3 factors that are impacting the balance of payment of your target country. 2. Forecast if the balance of payment would increase or decrease based on these factors in the next 12 months?
-
Consider the influence of digital technologies on work and change in organizations. Is technology a transforming solution to problems or a volatile disruption to work, organizational culture, and...
-
Conduct a macro-environment analysis for MYER. (Australian company)You should use the the PESTLE framework. focus is placed on the understanding of the purpose of this analysis the identification of...
-
Consider the following first order decompositions with rate constants as shown If Cs = 0.2 C A0 in the exit stream of a plug flow reactor, what are the concentration of the other reactant components...
-
Is times interest earned meaningful for utilities? Why or why not?
-
The equilibrium conversion is shown below as a function of catalyst weight down a PBR. The graph compares the values of conversion, X subscript e, and weight W. Initially at W equals 0, the...
-
The ethylene epoxydation is to be carried out using a cesium-doped silver catalyst in a packed-bed reactor. Along with the desired reaction, the complete combustion of ethylene also occurs It is...
-
Use the reaction data in Problem P6-11B and the molar flow rate algorithm to carry out the following problems: a. Calculate the CSTR volume to achieve 80% of the equilibrium conversion for an equal...
-
A dwarf planet moves around the Sun on an elliptical orbit with semi-major axis \(a\), semi-minor axis \(b\) and period of revolution \(T\). Determine the speed when its direction is perpendicular to...
-
Calculate the radius \(R_{S}\) of the orbit of a geostationary satellite using the lunar period, \(T=27.3\) d, and the Earth-Moon distance, \(D_{T L}=3.810^{5} \mathrm{~km}\), as data.
-
In the case of a gravitationally bound two-body system on an elliptical orbit in which there is energy dissipation as in the case of the question 4, explain why the system first tends to have a...

Study smarter with the SolutionInn App


