A. Standardization of Sodium Hydroxide (NaOH) Solution Trial 1 Mass of bottle + KHP Mass of...
Fantastic news! We've Found the answer you've been seeking!
Question:
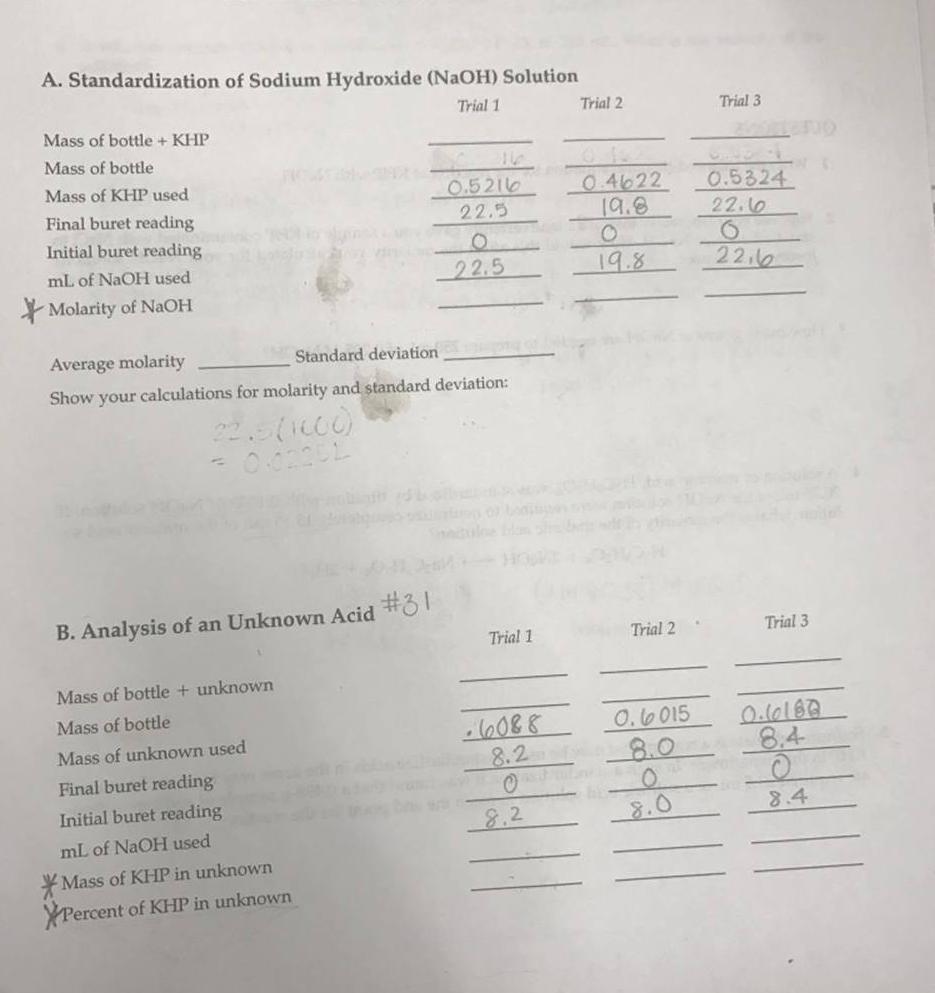
Transcribed Image Text:
A. Standardization of Sodium Hydroxide (NaOH) Solution Trial 1 Mass of bottle + KHP Mass of bottle Mass of KHP used Final buret reading Initial buret reading mL of NaOH used Molarity of NaOH B. Analysis of an Unknown Acid #31 Average molarity Standard deviation Show your calculations for molarity and standard deviation: 22.5(1000) 0.02.201 Mass of bottle + unknown Mass of bottle Mass of unknown used Final buret reading Initial buret reading mL of NaOH used 16 0.5216 22.5 *Mass of KHP in unknown Percent of KHP in unknown O 22.5 Trial 1 -6088 8.2 0 8.2 Trial 2 0.4622 19.8 O 19.8 Trial 2 0.6015 8.0 0 Trial 3 0.5324 22.6 6 22,6 Trial 3 0.60188 8.4 O 8.4 A. Standardization of Sodium Hydroxide (NaOH) Solution Trial 1 Mass of bottle + KHP Mass of bottle Mass of KHP used Final buret reading Initial buret reading mL of NaOH used Molarity of NaOH B. Analysis of an Unknown Acid #31 Average molarity Standard deviation Show your calculations for molarity and standard deviation: 22.5(1000) 0.02.201 Mass of bottle + unknown Mass of bottle Mass of unknown used Final buret reading Initial buret reading mL of NaOH used 16 0.5216 22.5 *Mass of KHP in unknown Percent of KHP in unknown O 22.5 Trial 1 -6088 8.2 0 8.2 Trial 2 0.4622 19.8 O 19.8 Trial 2 0.6015 8.0 0 Trial 3 0.5324 22.6 6 22,6 Trial 3 0.60188 8.4 O 8.4
Expert Answer:
Answer rating: 100% (QA)
For the analysis of the unknown acid The percent of KHP in the unknown c... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
A process for concentrating orange juice takes a feed of 100 kg/h, with a sugar mass percentage of 12%, and concentrates it to 45% using the flow diagram given below. A proportion of the feed...
-
An aqueous solution is 20.0% by mass of sodium thiosulfate pentahydrate, Na2S2O35H2O. What is the molarity of Na2S2O3 in this solution at 20C? The density of this solution at 20C is 1.174 g/mL.
-
A quantity of 0.25 M sodium hydroxide is added to a solution containing 0.15 mol of acetic acid. The final volume of the solution is 375 mL and the pH of this solution is 4.45. a. What is the molar...
-
Understand the content theories of motivation.
-
In India, a drug used to treat sick cows is leading to the death of many vultures that feed off of dead cattle. Before the decrease in the number of vultures, they sometimes used to smash into the...
-
Your department is one of the top producers in the company. Members work as a team without you. However, lately you realize there is a conflict within the group, but performance has not dropped due...
-
Which of the following statements about lift is false? a. Lift can be interpreted as how much more likely the antecedent and consequent itemsets occur together than if they were totally unrelated. b....
-
Principe Technology Solutions, Inc., completed the following transactions during August 2012, its first month of operations: Aug 1 Received cash of $48,000 and issued common stock. 2 Purchased...
-
Zack is using the Treynor and Black optimisation method. He has determined that the optimal weighting in the active portfolio is 76.19%. The active portfolio has a residual standard deviation of 5%...
-
Prevosti Farms and Sugarhouse pays its employees according to their job classification. The following employees make up Sugarhouse's staff: Employee Number Name and Address Payroll information...
-
15. The table below shows ages of 5 pupils with the mean age of 12.6 years.
-
Define and explain the difference between these terms: publics, shareholders, and target audiences.
-
Why is it particularly important for many out-of-home vehicles to be memorable?
-
What is the difference between IMC, advertising, and integrated brand communication (IBC)?
-
What is the use of television advertising ideally suited for?
-
How do scripts and storyboards work together to produce a television ad?
-
Create a strategy on how dollar general can decrease or protect itself from an identified Weakness or Threat in your SWOT Analysis.
-
In 1995 Miguel purchased a home for $130,000. In 2000 he sold it for $170,000 and immediately purchased another one for $180,000, which he sold in 2007 for $235,000. How much taxable capital gain, if...
-
Explain why each of the following sets of quantum numbers would not be permissible for an electron, according to the rules for quantum numbers. a. n = 1, l = 0, ml = 0, ms = +1 b. n = 1, l = 3, ml1 =...
-
Using enthalpies of formation (Appendix C), calculate Ho for the following reaction at 25oC. Also calculate So for this reaction from standard entropies at 25oC. Use these values to calculate Go for...
-
In 1986, Bernorz and Mller received the Nobel Prize in physics for what discovery? Why was this discovery important?
-
Derive the differential equation governing the motion of the one degree-of-freedom system by applying the appropriate form(s) of Newton's laws to the appropriate free-body diagrams. Use the...
-
Determine the differential equations governing the motion of the system by using the equivalent systems method. Use the generalized coordinates shown in Figures P2.52. k FIGURE P 2.52 E x 2k www
-
Determine the differential equations governing the motion of the system by using the equivalent systems method. Use the generalized coordinates shown in Figures P2.53. 2r k ww T m FIGURE P 2.53

Study smarter with the SolutionInn App


