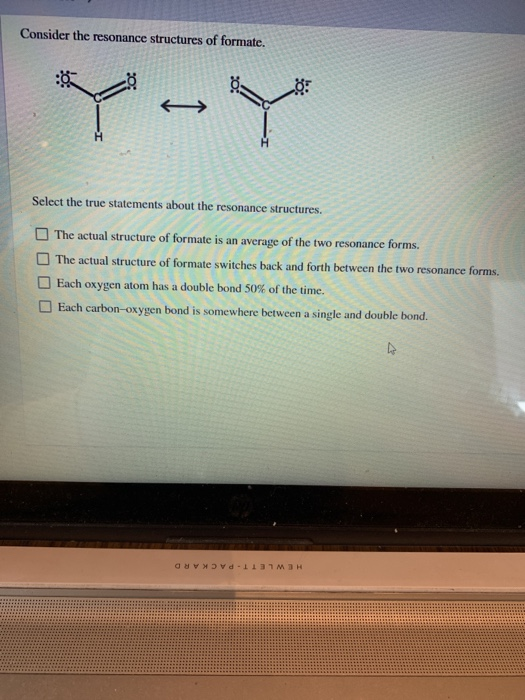
Consider the resonance structures of formate. Y-Y Select the true statements about the resonance structures. The...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the resonance structures of formate. Y-Y Select the true statements about the resonance structures. The actual structure of formate is an average of the two resonance forms. The actual structure of formate switches back and forth between the two resonance forms. Each oxygen atom has a double bond 50% of the time. Each carbon-oxygen bond is somewhere between a single and double bond. CONVd 113 Consider the resonance structures of formate. Y-Y Select the true statements about the resonance structures. The actual structure of formate is an average of the two resonance forms. The actual structure of formate switches back and forth between the two resonance forms. Each oxygen atom has a double bond 50% of the time. Each carbon-oxygen bond is somewhere between a single and double bond. CONVd 113
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Answer ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A dog runs back and forth between its two owners, who are walking toward one another (FIGURE 2-31). The dog starts running when the owners are 8.2 m apart. If the dog runs with a speed of 2.7 m/s,...
-
Consider the structure of nitrosobenzene: (a) Draw the resonance structures of the sigma complex formed when nitrosobenzene reacts with an electrophile (E+) at the ortho position. (b) Draw the...
-
The resonance structures of carbon monoxide are shown below. Show how each structure can be converted into the other using the curved-arrow notation. :C-: :C=0:
-
The annual membership fee at your health club is $750 a year and is expected to increase at 5% per year. A life membership is $7,500 and the discount rate is 12%. You either pay the annual membership...
-
On January 1, 2015, Water Mania issues $1,000,000 of 6% bonds, due in ten years, with interest payable semi-annually on June 30 and December 31 each year. Required: 1. If the market interest rate is...
-
Anstead Co. is experiencing a decrease in sales and operating income for the fiscal year ending October 31, 2014. Ryan Frazier, controller of Anstead Co., has suggested that all orders received...
-
After returning from a skiing vacation in Vermont, Leslie Adel came down with Legionnaires Disease. He claimed it was from the water drunk at the ski resort and provided by Greensprings of Vermont...
-
Gil Vogel started his own consulting firm, Vogel Consulting, on June 1, 2012. The trial balance at June 30 is as follows. In addition to those accounts listed on the trial balance, the chart of...
-
Schultz Incorporated ("Schultz") is a California manufacturer. Schultz employs several hourly, nonexempt workers in assembly-line positions. As a result, they are entitled to receive meal and rest...
-
Mark Sexton and Todd Story have been discussing the future of S&S Air. The company has been experiencing fast growth, and the two see only clear skies in the company's future. However, the fast...
-
Problem 3 (20 points) Derive the stress components for the following Airy stress function, plot the identified stress components on a rectangular element as shown, and speculate what problem the...
-
Non-marketable claims Hominy's debt has a face value of S215 million. If the company had raised capital solely from its net equity capital, its value would have been S11.4 million. The company also...
-
What Custom reports are used when standard reports do not yield the level of detail needed?
-
What are the tax implications of allocation methods? 10. How can you ensure the accuracy and fairness of cost allocations? How Estimates show that 4% of its receivables will be uncollectible?
-
How do ethical frameworks such as deontology, consequentialism, and virtue ethics inform decision-making in complex socio-technological environments?
-
Provide simple descriptions on the purpose of the Top brand & products report generated. The users of the Top brand & products report. How the Top brand & products report helps people in...
-
Using the information presented in Problem 13.4B, prepare a partial statement of cash flows for the current year, showing the computation of net cash flows from operating activities using the...
-
Which compound should undergo substitution of the bromine by phenolate anion: 4 - bromopyridine or 3 - bromopyridine ? Explain, and give the structure of the product.
-
Give the principal organic products expected when ethyl frarcs-2-butenoate (ethyl crotonate) reacts with each of the following reagents. (a) -CN in ethanol, then H2O/H3O+, heat (b) Me2NH, room...
-
AQC-typtoptan is not shown in Fig. 26.3, in what general region of the chromatogram would you expect to find AQC-Trp if it ware preseirr? Explain.
-
Let \(X_{1}, \ldots, X_{n}\) be a set of independent and identically distributed random variables from a distribution \(F\) with variance \(\mu_{2}\) where \(E\left(\left|X_{1}ight|^{4}ight) a. Prove...
-
Consider a sequence of independent random variables \(\left\{X_{n}ight\}_{n=1}^{\infty}\) where \(X_{n}\) has probability distribution function \[f_{n}(x)= \begin{cases}2^{-(n+1)} &...
-
Let \(X_{1}, \ldots, X_{n}\) be a set of independent and identically distributed random variables from a shifted exponential density of the form \[f(x)= \begin{cases}\exp [-(x-\theta)] & \text { for...

Study smarter with the SolutionInn App


