The water in a tank is pressurized by air, and the pressure is measured by a...
Fantastic news! We've Found the answer you've been seeking!
Question:
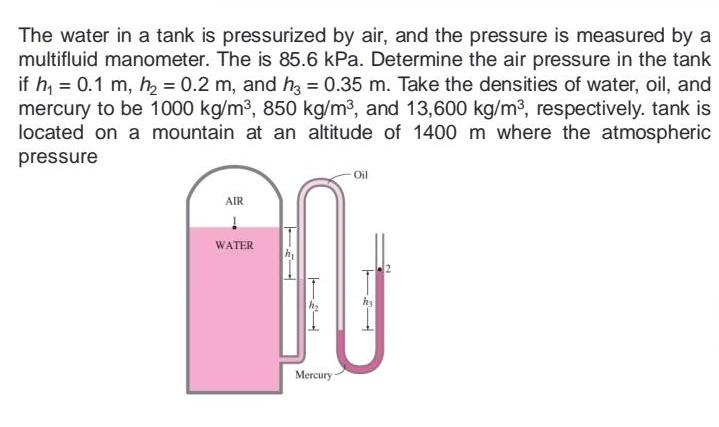
Transcribed Image Text:
The water in a tank is pressurized by air, and the pressure is measured by a multifluid manometer. The is 85.6 kPa. Determine the air pressure in the tank if h, = 0.1 m, h = 0.2 m, and hz = 0.35 m. Take the densities of water, oil, and mercury to be 1000 kg/m3, 850 kg/m3, and 13,600 kg/m3, respectively. tank is located on a mountain at an altitude of 1400 m where the atmospheric pressure Oil AIR WATER Mercury - The water in a tank is pressurized by air, and the pressure is measured by a multifluid manometer. The is 85.6 kPa. Determine the air pressure in the tank if h, = 0.1 m, h = 0.2 m, and hz = 0.35 m. Take the densities of water, oil, and mercury to be 1000 kg/m3, 850 kg/m3, and 13,600 kg/m3, respectively. tank is located on a mountain at an altitude of 1400 m where the atmospheric pressure Oil AIR WATER Mercury -
Expert Answer:

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these chemical engineering questions
-
The water in a tank is pressurized by air, and the pressure is measured by a multi fluid manometer as shown in Fig. P142 Determine the gage pressure of air in the tank if h1 = 0.2 m, h2 = 0.3 m, and...
-
The water in a tank is pressurized by air, and the pressure is measured by a multifluid manometer as shown in Fig. P1-50. Determine the gage pressure of air in the tank if h1 = 0.2 m, h2 = 0.3 m, and...
-
The pressure in a container on a mountain at an elevation of 25,000 ft is measured to be a) 5 psi b) 20 psi. What is the absolute pressure in psia? In psfa?
-
Compare sand, die, investment, lost foam, and continuous casting techniques.
-
Suppose the price of regular-octane gasoline were 20 cents per gallon higher in New Jersey than in Oklahoma. Do you think there would be an opportunity for arbitrage (i.e., that firms could buy gas...
-
Rainbow Glass Company manufactures decorative glass products. The firm employs a process-costing system for its manufacturing operations. All direct materials are added at the beginning of the...
-
Consider the calculation of an external rate of return (ERR). The positive cash flows in the cash flow profile are moved forward to \(t=n\) using what value of \(i\) in the \((F \mid P i, n-t)\)...
-
Ross Textiles wishes to measure its cost of common stock equity. The firms stock is currently selling for $57.50. The firm expects to pay a $3.40 dividend at the end of the year (2013). The dividends...
-
If V f(xz, y/z), prove that zV =xVx-yVy. =
-
Mike Curtains, a Registered Tax Agent, attends to the tax affairs of Frodo West. In preparing Frodo's 2021/22 income tax return, the following occurred: Frodo attended a meeting with Mike in August...
-
Rainbow Company sells products that are subject to a 1-year warranty. During 2016, the company sold 70,000 units of its products, of which management believes that 5% of the units will be defective....
-
Retro Gym has three creditors: a bank with seniority in the amount of $350, Senior Bondholders in the amount of $190, and a steel trade creditor with no priority in the amount of $40. Retro Gym has...
-
Best Cakes requires a certain part for its baking oven to continue to stay open. Best Cakes orders this part from Oven Repairs Inc. Best Cakes tells Oven Repairs that is must get the item by Friday...
-
Brooklyn Borough Hospital is examining its inpatient services to determine the year-to-year change in revenue. After examining the chart below, please answer the questions listed. 2022 Revenue...
-
Individual Social Media Analysis Assignment: This week, each group member will select a different social media platform as the subject for an individual social media assignment. I am looking for ...
-
Context diagram shows the flow of information constraints who and what is doing the work all the above Question 5 (1 point) BUC's are responses to business events True False Question 6 (1 point)...
-
1. Caitlin, Chris, and Molly are partners and share income and losses in a 3:4:3 ratio. The partnerships capital balances are Caitlin, $128,000; Chris, $88,000; and Molly, $108,000. Paul is admitted...
-
Q1) What is the a3 Value Q2) What is the a7 Value Q3) What is the a4 Value Q4) What is the b3 Value Q5) What is the b2 Value Q6) What is the sign of 2nd constraint? A pastry chef at a bakery wants to...
-
The equation of state of a gas is given as v-(P + 10/ v-2) = 5 RuT, where the units of v- and P are m3/kmol and kPa, respectively. Now 0.2 kmol of this gas is expanded in a quasi equilibrium manner...
-
In a heating system, cold outdoor air at 7C flowing at a rate of 4 kg/min is mixed adiabatically with heated air at 70C flowing at a rate of 3 kg/min. The exit temperature of the mixture is (a) 34C...
-
One pound-mass of water fills a 2.649 ft3 weighted piston-cylinder device at a temperature of 400oF. The piston-cylinder device is now cooled until its temperature is 100oF. Determine the final...
-
Stadler Corporations federal income tax rate is 34 percent. It reports $100,000 depreciation expense on its financial statements and deducts $140,000 depreciation expense on its tax return. How...
-
Which of the following items is not deductible? a. Dues for club used solely for business meetings b. Directly related business entertainment c. Business gift of less than $25 in value d. Dues for...
-
John is a teacher at a local high school. During 2017, he travels three days per week to a school in the next county to work with gifted children in an after-school program that does not end until...

Study smarter with the SolutionInn App


