Explain why the first ionization energy of Ca is greater than that of K, whereas the...
Fantastic news! We've Found the answer you've been seeking!
Question:
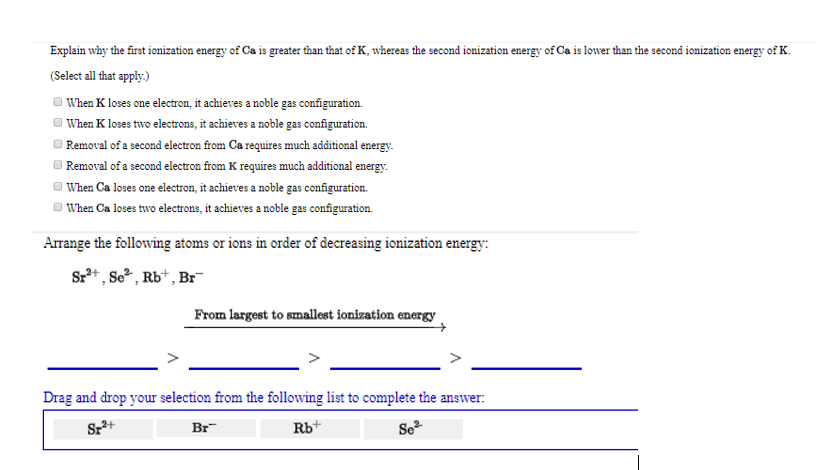
Transcribed Image Text:
Explain why the first ionization energy of Ca is greater than that of K, whereas the second ionization energy of Ca is lower than the second ionization energy of K. (Select all that apply.) When K loses one electron, it achieves a noble gas configuration. When K loses two electrons, it achieves a noble gas configuration. Removal of a second electron from Ca requires much additional energy. Removal of a second electron from K requires much additional energy. When Ca loses one electron, it achieves a noble gas configuration. When Ca loses two electrons, it achieves a noble gas configuration. Arrange the following atoms or ions in order of decreasing ionization energy: Sr²+, So², Rb+, Br From largest to smallest ionization energy Drag and drop your selection from the following list to complete the answer: Sz²+ Br Rb+ Se² Explain why the first ionization energy of Ca is greater than that of K, whereas the second ionization energy of Ca is lower than the second ionization energy of K. (Select all that apply.) When K loses one electron, it achieves a noble gas configuration. When K loses two electrons, it achieves a noble gas configuration. Removal of a second electron from Ca requires much additional energy. Removal of a second electron from K requires much additional energy. When Ca loses one electron, it achieves a noble gas configuration. When Ca loses two electrons, it achieves a noble gas configuration. Arrange the following atoms or ions in order of decreasing ionization energy: Sr²+, So², Rb+, Br From largest to smallest ionization energy Drag and drop your selection from the following list to complete the answer: Sz²+ Br Rb+ Se²
Expert Answer:
Answer rating: 100% (QA)
Answer 1 The first ionization energy of an element is the energy required to remove one electron from a neutral atom of that element Calcium has an at... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Explain why the first ionization energy tends to increase as one proceeds from left to right across a period. Why is the first ionization energy of aluminum lower than that of magnesium and the first...
-
From Figure 8.18, predict the first ionization energy of ununseptium (Z = 117). 2500 He 2000 * 1500 Kr Br As Rn E 1000 Bi S00 Li Ca Sr Na In Ba K Rb Cs 10 18 36 54 86 Atomic number, Z -Ne 550 1008...
-
From Figure 8.18, predict the first ionization energy of francium (Z = 87). 2500 He 2000 * 1500 Kr Br As Rn E 1000 Bi S00 Li Ca Sr Na In Ba K Rb Cs 10 18 36 54 86 Atomic number, Z -Ne 550 1008 At...
-
Ebrahim Patel is a wholesaler who uses the periodic inventory system to account for inventory. Transactions for February: 1 Bought inventory from Rich Traders for R5 000 on credit. 2 Sold inventory...
-
Compute Paasches index for 2013 using 2000 as the baseperiod. Price Quantity Item Cabbage (pound $0.06 $0.05 2,000 1,500 Carrots (bunch) Peas (quart) Endive (bunch) 2000 2013 2000 2013 0.10 0.20 0.15...
-
Which rock should be most resistant to chemical weatheringgabbro or granite?
-
Xie Company identified the following activities, costs, and activity drivers for 2017. The company manufactures two types of go-karts: deluxe and basic. Required 1. Compute a single plantwide...
-
The S&P stock index represents a portfolio comprised of 500 large publicly traded companies. On December 24, 2007, the index had a value of 1,410 and on December 23, 2008, the index was approximately...
-
Thunderduck Shoes provides shoe shining and repair services to customers. For the year which ended Dec 31, the company reports the following amounts: Account Amount Account Amount Rent Expense 22,400...
-
Effective financial statement analysis requires an understanding of a firms economic characteristics. The relations among various financial statement items provide evidence of many of these economic...
-
Discuss the relationship Matrix and how companies prioritize some relationships over others. How will this impact the future of how the supply chain operates? Examine what motivates businesses to...
-
What can a business do to improve its productivity? Explain how ?
-
if the atomic radius of a cubic monatomic crystal with FCC unit cell is 0 , 1 5 0 nm , how many atoms are there per square nanometer of planes with ( 1 1 0 ) miller index ? Briefly explain with...
-
Suppose the following order arrive in sequence: 1. Harry: Buy 700 stocks at market. 2. Jenny: Sell 1600 stocks at limit $50.04. What is the bid ask spread after these orders arrive and are executed?...
-
Estate Tax Planning GOALS FOR SYNC SESSION Review of some Estate Tax Planning Strategies In-Class Practice - Taxable gift? Practice Problems with Estate Taxes Questions about Exam 3 Review of some...
-
The Sun moves in a circular orbit around our Milky Way galaxy with an orbital radius r = 2.5x10 20 m m and a uniform circular speed v = 2x10 5 m/s. Estimate the mass of the galaxy inside the Sun...
-
On August 1, 2014, Larry Goldstein and Rafi Hassan created a partnership to produce software for online advertising. Goldstein was a lawyer and would handle all the legal matters, but Hassan was the...
-
The following cost information was provided to you for analysis: September 12,000 Units Produced Costs: TIC TAC TOE TING August 10,000 P80,000 70.000 60.000 50,000 How much is the fixed cost per...
-
Draw cyclic structures for D-ribose and D-mannose.
-
Balance the following equations. a. Cr(s) + S8(s) ( Cr2S3(s) Heat b. NaHCO3(s) ( Na2CO3(s) + CO2(g) + H2O(g) Heat c. KClO3(s) ( KCl(s) + O2(g) d. Eu(s) + HF(g) ( EuF3(s) + H2(g) e. C6H6(l) + O2(g) (...
-
What is the valence electron configuration for alkaline earth metals? List some common properties of alkaline earth metals. How are alkaline earth metals prepared?
-
Taylor Pty Ltd is based in Brisbane and sells glamping yurt tents imported from China at a cost of $150 each plus import duties and other costs of $60. These purchase and import costs are paid cash...
-
ArbreCorp Ltee is a sawmill operation in Quebec that is owned by a major forest products company but operates independently of the parent company. It was built 30 years ago, and completely updated...
-
Over the past 15 years, Volkswagen Group (VW) acquired several fiefdoms- Audi, Lamborghini, Bentley, Bugatti, Skoda, SEAT - that jealously guarded their brand and continuously rebelled against...

Study smarter with the SolutionInn App


