Question: For compounds A and B shown below, provide the reaction conditions needed to remove the protecting group and draw the resulting deprotected molecule. For
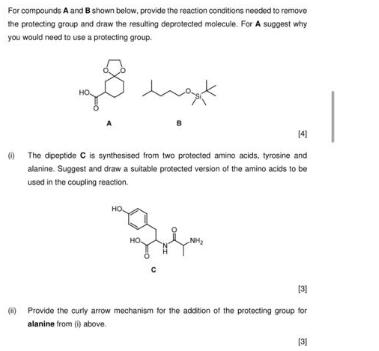
For compounds A and B shown below, provide the reaction conditions needed to remove the protecting group and draw the resulting deprotected molecule. For A suggest why you would need to use a protecting group. want 141 (The dipeptide C is synthesised from two protected amino acids, tyrosine and alanine. Suggest and draw a suitable protected version of the amino acids to be used in the coupling reaction. HO HO NH [3] Provide the curly arrow mechanism for the addition of the protecting group for alanine from () above (31
Step by Step Solution
3.45 Rating (161 Votes )
There are 3 Steps involved in it
Step 12 a For compound A a protecting group is needed because the amino group is a nucleophile and c... View full answer

Get step-by-step solutions from verified subject matter experts


