k1 gt) What would be the major organic product of the following reaction? CH3 H HC-C-C...
Fantastic news! We've Found the answer you've been seeking!
Question:

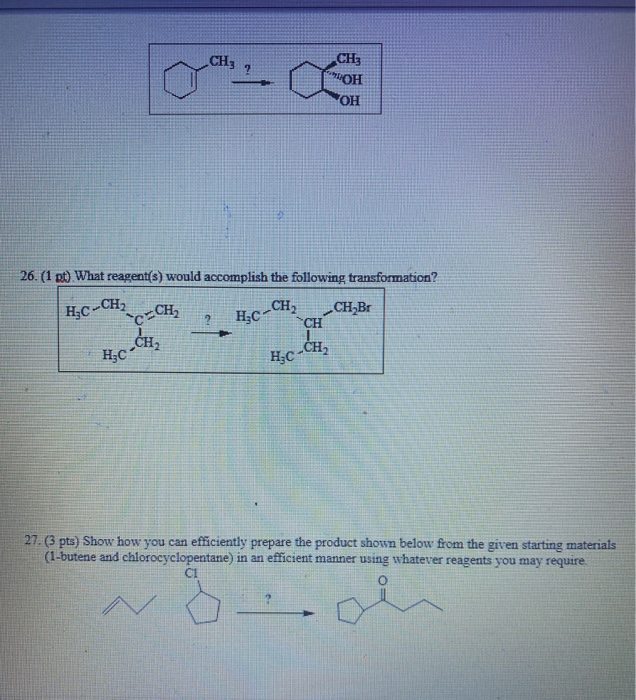
Transcribed Image Text:
k1 gt) What would be the major organic product of the following reaction? CH3 H HC-C-C CH₂ CH₂ HCI 2CH₂ CH₂ 2 24. (1 pt) What would be the major organic product of the following reaction? H₂C-CH₂-CH₂ H₂O.H₂SO4 2 + 25. (1 pt) What set of reagents would convert 1-methylcyclohexene into trans-1-methyl-1,2- cyclohexanediol? H₂C- H₂C CH3 26. (1 pt) What reagent(s) would accomplish the following transformation? -CH₂ H₂C-CH₂ C-CH₂ ? ? CH H₂C-CH₂ CH3 OH OH ➤ CH₂Br 27. (3 pts) Show how you can efficiently prepare the product shown below from the given starting materials (1-butene and chlorocyclopentane) in an efficient manner using whatever reagents you may require. CI N k1 gt) What would be the major organic product of the following reaction? CH3 H HC-C-C CH₂ CH₂ HCI 2CH₂ CH₂ 2 24. (1 pt) What would be the major organic product of the following reaction? H₂C-CH₂-CH₂ H₂O.H₂SO4 2 + 25. (1 pt) What set of reagents would convert 1-methylcyclohexene into trans-1-methyl-1,2- cyclohexanediol? H₂C- H₂C CH3 26. (1 pt) What reagent(s) would accomplish the following transformation? -CH₂ H₂C-CH₂ C-CH₂ ? ? CH H₂C-CH₂ CH3 OH OH ➤ CH₂Br 27. (3 pts) Show how you can efficiently prepare the product shown below from the given starting materials (1-butene and chlorocyclopentane) in an efficient manner using whatever reagents you may require. CI N
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Help & Save is a private not-for-profit organization that operates in Kansas. Swim For Safety is a private not-for-profit organization that operates in Missouri. The leaders of these two...
-
Leadership skills can help you in all aspects of your career, from applying for jobs to seeking career advancement. One of the many soft skills that employers value, leadership often incorporates...
-
Help & Save is a private not-for-profit entity that operates in Kansas. Swim For Safety is a private not-for-profit entity that operates in Missouri. The leaders of these two organizations have...
-
You are given the following as it relates to the price of a pound of flour in Adam Island, 2016 to 2022: 2016-$1.35 2017 - $1.50 2018 - $1.65 2019 - $1.85 2020 - $2.15 2021 - $2.00 2022 - $2.10 Using...
-
Vitron Inc. is considering an investment in new capital equipment that will cost 250,000 plus an additional 15,000 investment in inventory to operate the equipment. They expect sales to increase by...
-
Determine the absolute maximum shear in the bridge girder due to the loading shown. 10 k 6k 8 ft B 30 ft-
-
Pluto Corporation issued \(\$ 400,000\) of \(7 \%, 20\) year bonds payable on March 31, 2006. The bonds were issued at 100 and pay interest on March 31 and September 30. Record (a) issuance of the...
-
At the end of the year, Email Enterprises estimates the uncollectible accounts expense to be 0.8 percent of net sales of $7,575,000. The current credit balance of Allowance for Uncollectible Accounts...
-
Question1 Motivationis agoal-oriented characteristicthat helpsapersonachievehis objectives. It pushesanindividualtoworkhardatachievinghisorhergoals.Anexecutivemusthavethe...
-
SAE specifications call for the low-side R-134a servicehose to be A) Solid blue with a black stripe B) Solid blue with no stripe C) Solid blue with a yellow stripe D) Solid black with a blue stripe...
-
Synthesis Problem: 6. Consider a chemical reaction where A- as: (a) Use na, n, nc to represent the populations of A, B, and C. Write down the coupled rate equations describing the populations of A,...
-
The table below represents the market for bushels of wheat. Suppose there is a price floor set at $15 for a bushel of wheat. Calculate the surplus caused by the price floor. Price Quantity Supplied...
-
What is the hydroxide ion concentration and the pH of a solution that has a hydronium ion concentration of 4.50 10-
-
eBook Bank Reconciliation Instructions Amount Descriptions Chart Of Accounts Amount Descriptions Bank Reconciliation Instructions Show Me How 2 Valentine Investigations has the following information...
-
An atmospheric drag force with magnitude FD=Dv2 , where v is speed, acts on a falling 360 mg raindrop that reaches a terminal velocity of 12 m/s .Find the value of D .
-
A survey of a local market has provided the following average cost data: Johnson Construction Corporation (JCC) has assets of $3 million and an average cost of 19 percent. Anderson Architects (AA)...
-
Objective: Execute the actual change, moving manufacturing to the new location while maintaining operational continuity. Implementation Plan: Phased Transition: Implement the change in phases,...
-
Which property determines whether a control is available to the user during run time? a. Available b. Enabled c. Unavailable d. Disabled
-
Rank the following anions in order of decreasing basicity: CH O CH,o CH
-
Which of the following hexachlorocyclohexanes is the least reactive in an E2 reaction? CI CI CI Cl Ci C Cl, Cl, Cl C CI Cl CT Cl C Cl Cl CI CI CI
-
cis-3,4-Dimethylcyclobutene undergoes thermal ring opening to form the two products shown. One of the products is formed in 99% yield, the other in 1% yield. Which is which? CH CH
-
Consider the following statements: Radius of friction circle for a journal bearing depends upon 1). coefficient of friction 2). radius of the journal 3). angular speed of rotation of the shaft Which...
-
Consider the following statements: 1). A stub tooth has a working depth larger than that of a full-depth tooth. 2). The path of contact for involute gears in an arc of a circle. Which of the...
-
Consider the following statements: Cam followers are generally classified according to 1). the nature of its motion 2). the nature of its surface in contact with the cam 3). the speed of the cam...

Study smarter with the SolutionInn App


