Highly charged metal ions are often strong acids. Now let me explain why it is so. Obviously,
Fantastic news! We've Found the answer you've been seeking!
Question:
Highly charged metal ions are often strong acids. Now let me explain why it is so. Obviously, in aqueous solution we have water and oxygen of water is partially negative:

As a result water forms complex with the metal ions. For example, Fe+3 will form hexa coordinated complex as following:

Thus, metal ions (with strong positive charge) in aqueous solution favors the following equilibrium. Below is an example:
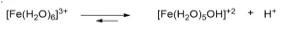
As a consequence the pH of the [Fe(H₂O)]³+ even goes down to 1.5
Related Book For 

Advertising and Promotion An Integrated Marketing Communications Perspective
ISBN: 978-0078028977
10th Edition
Authors: George Belch, Michael Belch
Posted Date:




