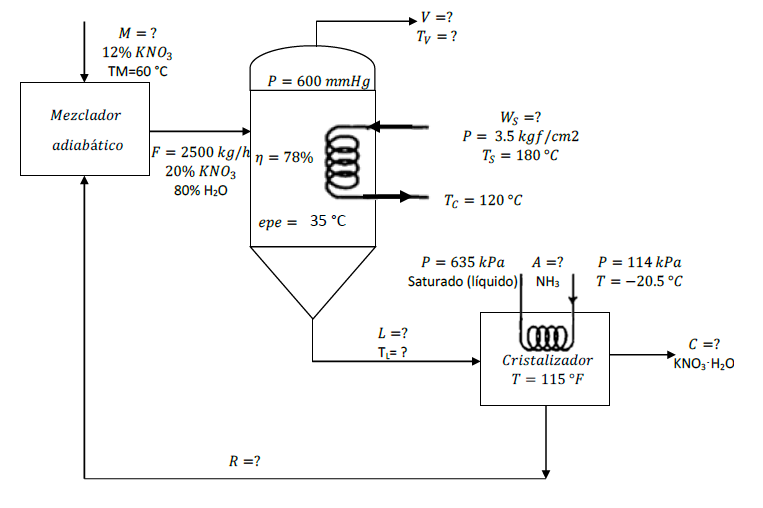
I. In a process that produces KNO3 ? H2O (potassium nitratemonohydrate), the evaporator is fed with 2500
Fantastic news! We've Found the answer you've been seeking!
Question:
I. In a process that produces KNO3 ? H2O (potassium nitratemonohydrate), the evaporator is fed with 2500 kg / h of a solutioncontaining 20% KNO3 by weight and is concentrated at a pressure of600 mmHg, obtaining an increase in boiling point of 35 ? C. Thisconcentrated solution is fed to a crystallizer that works at 115 ?F, where KNO3 ? H2O crystals are obtained, the mother liquorsolution, which contains 37.5% weight of KNO3, is recirculated tothe evaporator. Calculate the required flows, if the heat transferefficiency in the evaporator is 78%. Neglect the heat ofmixing.
Related Book For 

Managing Controlling and Improving Quality
ISBN: 978-0471697916
1st edition
Authors: Douglas C. Montgomery, Cheryl L. Jennings, Michele E. Pfund
Posted Date:




