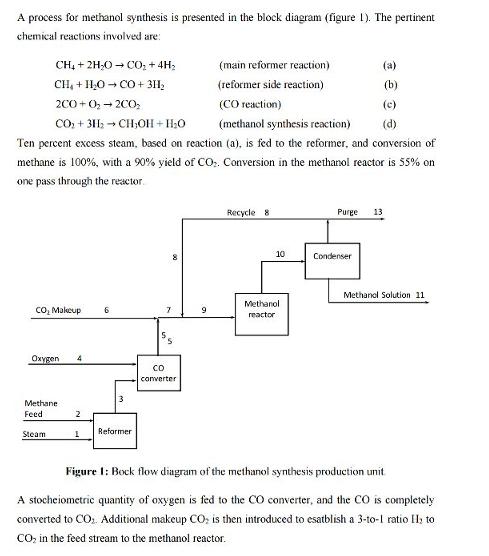
A process for methanol synthesis is presented in the block diagram (figure 1). The pertinent chemical...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
A process for methanol synthesis is presented in the block diagram (figure 1). The pertinent chemical reactions involved are: CH₂ + 2H₂O → CO₂ + 4H₂ CH₂ + H₂O →CO + 3H₂ 2C0+0₂ - 200₂ CO₂ + 3H₂CH₂OH+H₂O (methanol synthesis reaction) (d) Ten percent excess steam, based on reaction (a), is fed to the reformer, and conversion of methane is 100%, with a 90% yield of CO₂. Conversion in the methanol reactor is 55% on one pass through the reactor. CO₂ Makeup 6 Oxygen 4 Methane Feed Steam 2 1 3 Reformen CO converter (main reformer reaction) (reformer side reaction) (CO reaction) 9 Recycle 81 10 Methanol reactor (a) (b) (c) Purge 13 Condenser Methanol Solution 11 Figure 1: Bock flow diagram of the methanol synthesis production unit A stocheiometric quantity of oxygen is fed to the CO converter, and the CO is completely converted to CO₂. Additional makeup CO₂ is then introduced to esatblish a 3-to-1 ratio 1₂ to CO₂ in the feed stream to the methanol reactor. 2| Page The methanol reactor effluent is cooled to condense all the methanol and water, with the noncondensible gases recycled to the methanol reactor feed. The H/CO₂ ratio in the recylee stream is also 3-to-1. Because the methane feed contains 1% nitrogen as an impurity, a portion of the receycle stream must be purged as shown in the figure I to prevent the accumulation of nitrogen in the system. The purge stream analyzes 5% nitrogen. 1) Sketch the PFD f the process and label all the streams, blocks etc... according to the guidelines presented in "Lecture_02_bonus_GCH350", 2) On the basis of 100 mol/h of methane feed (inculding the N₂), calculate: a. How many moles of H₂ are lost in the purge. b. How many moles of makeup CO₂ are required. c. The recycle to purge ratio in mol/mol. d. How much methanol solution (in kg/h) of what strength (weight percent) is produced. 3 | Page A process for methanol synthesis is presented in the block diagram (figure 1). The pertinent chemical reactions involved are: CH₂ + 2H₂O → CO₂ + 4H₂ CH₂ + H₂O →CO + 3H₂ 2C0+0₂ - 200₂ CO₂ + 3H₂CH₂OH+H₂O (methanol synthesis reaction) (d) Ten percent excess steam, based on reaction (a), is fed to the reformer, and conversion of methane is 100%, with a 90% yield of CO₂. Conversion in the methanol reactor is 55% on one pass through the reactor. CO₂ Makeup 6 Oxygen 4 Methane Feed Steam 2 1 3 Reformen CO converter (main reformer reaction) (reformer side reaction) (CO reaction) 9 Recycle 81 10 Methanol reactor (a) (b) (c) Purge 13 Condenser Methanol Solution 11 Figure 1: Bock flow diagram of the methanol synthesis production unit A stocheiometric quantity of oxygen is fed to the CO converter, and the CO is completely converted to CO₂. Additional makeup CO₂ is then introduced to esatblish a 3-to-1 ratio 1₂ to CO₂ in the feed stream to the methanol reactor. 2| Page The methanol reactor effluent is cooled to condense all the methanol and water, with the noncondensible gases recycled to the methanol reactor feed. The H/CO₂ ratio in the recylee stream is also 3-to-1. Because the methane feed contains 1% nitrogen as an impurity, a portion of the receycle stream must be purged as shown in the figure I to prevent the accumulation of nitrogen in the system. The purge stream analyzes 5% nitrogen. 1) Sketch the PFD f the process and label all the streams, blocks etc... according to the guidelines presented in "Lecture_02_bonus_GCH350", 2) On the basis of 100 mol/h of methane feed (inculding the N₂), calculate: a. How many moles of H₂ are lost in the purge. b. How many moles of makeup CO₂ are required. c. The recycle to purge ratio in mol/mol. d. How much methanol solution (in kg/h) of what strength (weight percent) is produced. 3 | Page
Expert Answer:
Answer rating: 100% (QA)
100mol feed into the reformer 99 mol Methane N2 1 mol Reactions 2H2O CO2 4H2 CH4 main CH4 CO2 3H2 si... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these chemical engineering questions
-
Determine the amount of heat removed from the reactor per mole of CH4 fed to the reactor. 2. Methane and oxygen at 25C are fed to a continuous reactor in stoichiometric amounts according to the...
-
A steady stream of equimolar N2 and CO2 mixture at 100 kPa and 18C is to be separated into N2 and CO2 gases at 100 kPa and 18C. Determine the minimum work required per unit mass of mixture to...
-
A light-hydrocarbon feed stream contains 45.4 kmol/h of propane, 136.1 kmol/h of isobutane, 226.8 kmol/h of n-butane, 181.4 kmol/h of isopentane, and 317.4 kmol/h of n-pentane. This stream is to be...
-
Carbon has the electron configuration 1s 2s2p. The two unpaired electrons in the n = 2 level suggests that carbon will form two bonds. We know, however, that carbon forms four bonds in most compounds...
-
An assembly of two parts is formed by fitting a shaft into a bearing. It is known that the inside diameters of bearings are normally distributed with mean 2.010 cm and standard deviation 0.002 cm,...
-
Find the tension T in each cable and the magnitude and direction of the force exerted on the strut by the pivot in each of the arrangements in Fig. 11.26. In each case let w be the weight of the...
-
What factors should be considered in setting alpha risk? How is this risk controlled?
-
Ramanan, Inc., has performed cost studies and projected the following annual costs based on 200,000 units of production and sales: Total Annual Costs (200,000 units) Direct Material...
-
Bronte Confections is known for its rich dark chocolate fudge. Bronte sells its fudge to local retailers. A "unit" of fudge is a 10-pound batch. The standard quantities of ingredients for a batch...
-
Goldie and Kurt want advice from your financial advising firm. They have provided the following information. They graduated from university four years ago and they have good jobs, but neither of them...
-
Andrew is contemplating between two three-year contract job opportunities. His first option guarantees him $100,000 per year, while his second option offers him $50,000 + a bonus per year. Suppose...
-
What are people usually motivated to do when they experience a face-threatening act?
-
In what ways can you categorize your key abilities and attributes to project credibility?
-
Under what circumstances is using powerless speech advantageous?
-
What are some ways you might undersell yourself on rsums?
-
Which forms of language are characterized as powerless?
-
Consider the following function: f(x)=x516.05x4 + 88.75x-192.0375x + 116.35x + 31.6875 Find the roof to f f(x) using (a) Newton-Raphson method and b) using the modified secant method (8=0.05). Use...
-
Use a calculator to evaluate the expression. Round your result to the nearest thousandth. V (32 + #)
-
Derive expressions for (a) u. (b) h. (c) s for a gas whose equation of state is P(v - a) = RT for an isothermal process.
-
Describe the solar radiation properties of a window that is ideally suited for minimizing the air-conditioning load.
-
Reconsider the scram jet engine discussed in Prob. 17-41. Determine the speed of this engine in miles per hour corresponding to a Mach number of 7 in air at a temperature of 00F.
-
A motorcycle travels over a road whose contour is approximately sinusoidal, \(y(z)=0.2 \sin (0.4 z) \mathrm{m}\) where \(z\) is measured in meters. Using a SDOF model, design a suspension system with...
-
A \(20 \mathrm{~kg}\) block is connected to a spring of stiffness \(1 \times 10^{5} \mathrm{~N} / \mathrm{m}\) and placed on a surface which makes an angle of \(30^{\circ}\) with the horizontal. A...
-
When a free-vibration test is run on the system of Figure P4.76, the ratio of amplitudes on successive cycles is 2.8 to 1 . When operating, the engine has a rotating unbalance of magnitude \(0.25...

Study smarter with the SolutionInn App


