Question: Methane gas is to be liquefied using the process shown in Figure Q1.1. The methane feed to the compressor is at 280 K and



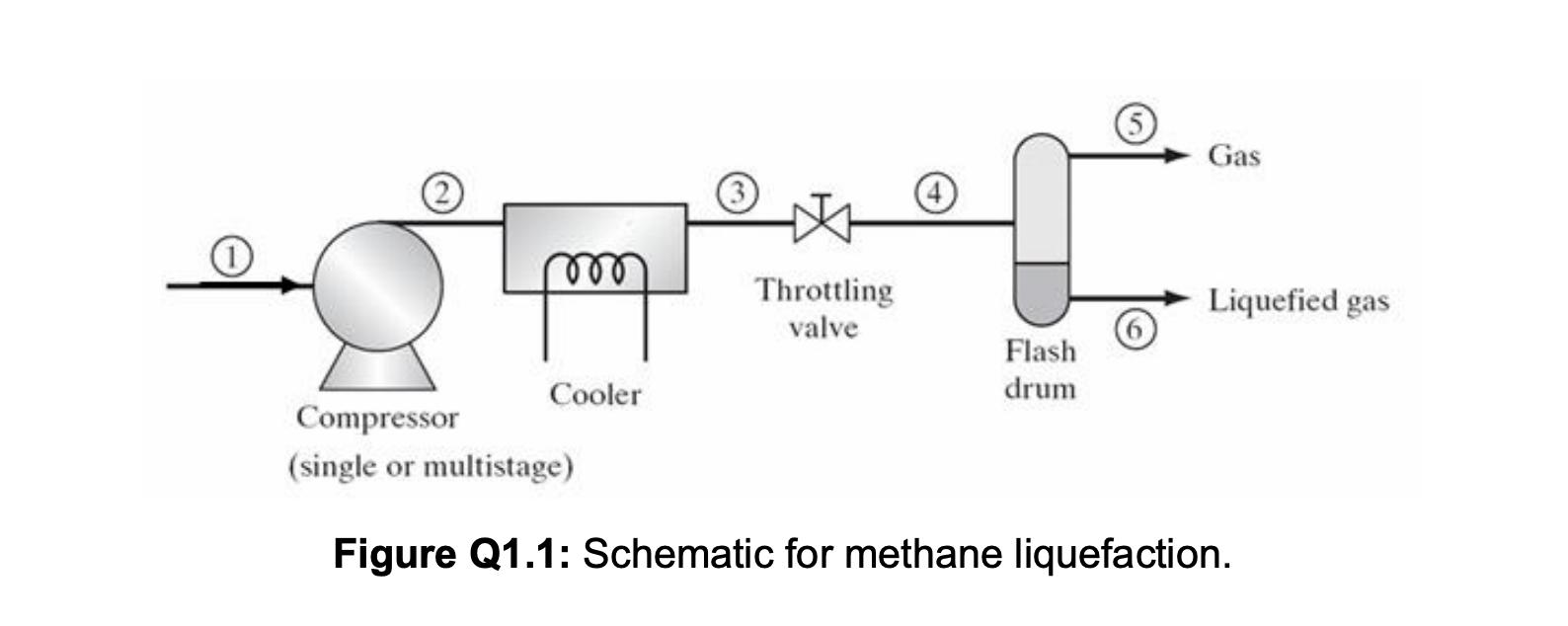
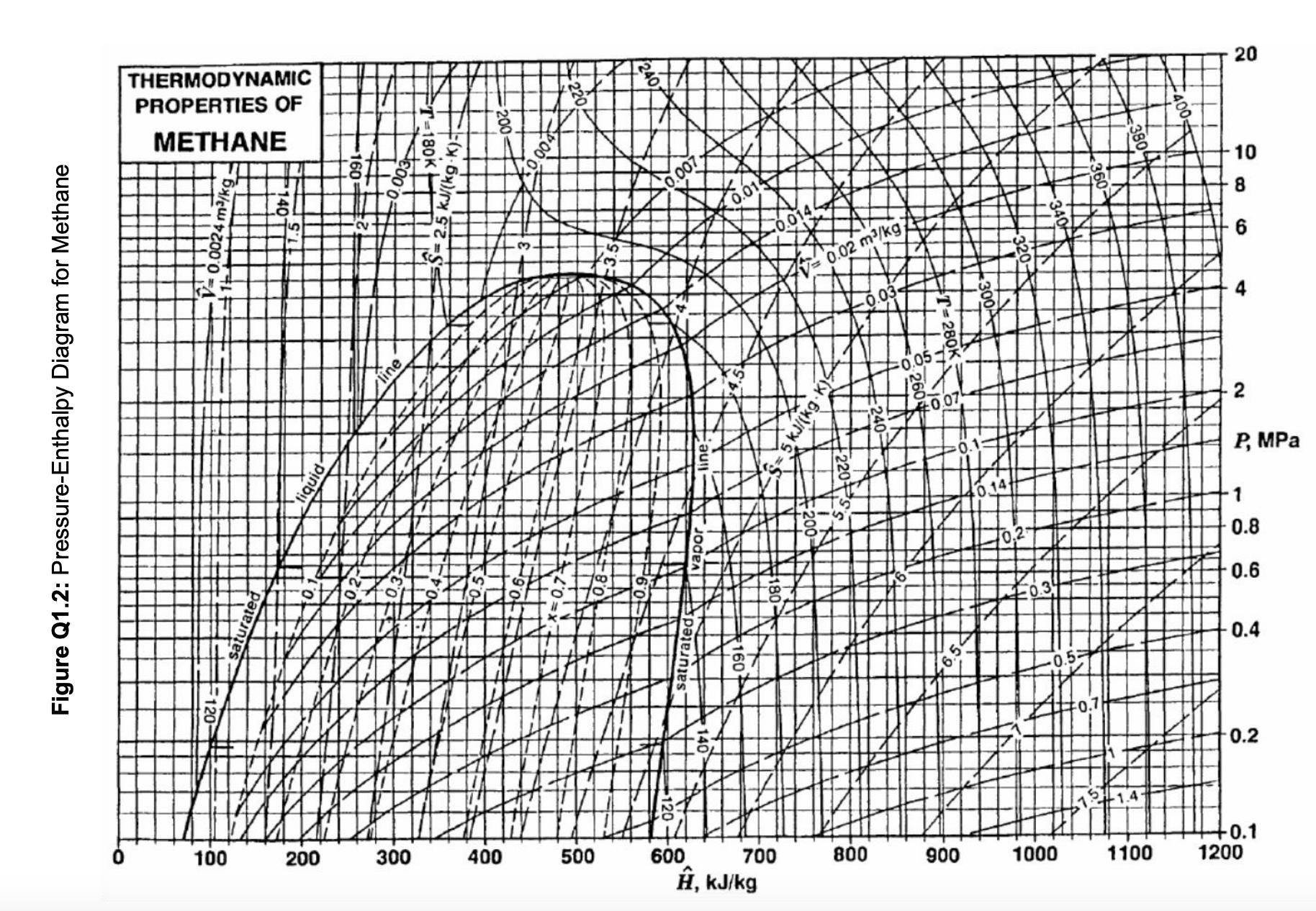
Methane gas is to be liquefied using the process shown in Figure Q1.1. The methane feed to the compressor is at 280 K and 1 bar, and compression takes place adiabatically and reversibly. After passing through a cooler, assumed to operate at constant pressure, methane is throttled to produce liquefied methane, which is subsequently separated in a flash drum where the pressure is 1 bar. a. For the process described above, devise a multistage compressor with isobaric intercooling between each compression stage, assuming the maximum compressor work available per kg of methane is 750 kJ and the total compressor intercooling capacity available is 500 kJ per kg of methane. Report (i) the pressure and temperature of methane leaving the compressor, (ii) the number of compression and intercooling stages, (iii) the work term for each compression stage, and (iv) the amount of heat removed during each intercooling step. Show the compression and intercooling stages in your work on the pressure-enthalpy diagram for methane provided in Figure Q1.2. b. Once methane leaves the compressor, it enters the cooler (shown between points 2 and 3 in Figure Q1.1 the figure above) before being throttled. Plot the liquid yield (i.e. fraction of methane liquefied) vs cooling duty (i.e. heat removed) for the cooler. You may use the pressure-enthalpy diagram for methane provided in Figure Q1.2. Compressor Cooler Throttling valve Gas Liquefied gas Flash drum (single or multistage) Figure Q1.1: Schematic for methane liquefaction. Figure Q1.2: Pressure-Enthalpy Diagram for Methane THERMODYNAMIC PROPERTIES OF METHANE 5 fiquid line 180K 2 KJ/(k . 0.004 240 0.007 0.014 0.0144 V= 0.02 m/kg) 0.03 S=5 kJ/kgK) 0.051x 0.07 0.1 0.14 0,2 10.34 +0.5-7 2 4 10 8 6 P, MPa 880 1 0.8 0.6 0.4 0.2 +0.1 100 200 300 400 500 600 700 800 900 1000 1100 1200 , kJ/kg -1.4- 20
Step by Step Solution
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


