P02. (30 pts) A two phase liquid-vapor mixture of HO, initially at equilibrium in state 1...
Fantastic news! We've Found the answer you've been seeking!
Question:

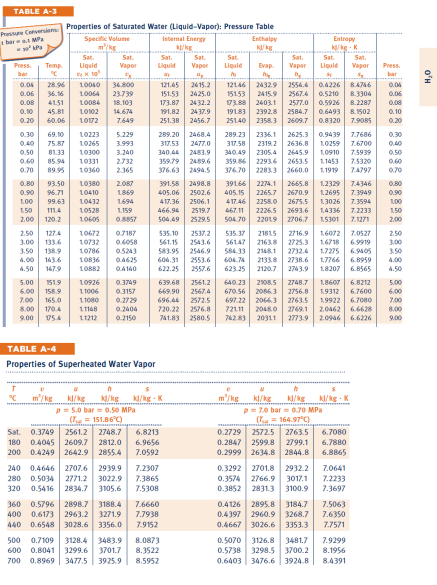
Transcribed Image Text:
P02. (30 pts) A two phase liquid-vapor mixture of HO, initially at equilibrium in state 1 with p = 1.5bar (150 kPa) with a quality of x = 75% is contained in a sealed tank with rigid walls. The mass of HO is m = 5 kg. a) (5) Determine the volume of the tank, V, in m. b) (5) Determine the total internal energy of the liquid-vapor mixture at state 1 Next, heat is added to the tank until the phase of the water in the tank becomes state 2, the saturated vapor state. c) (5) determine the pressure of the state 2 d) (5) Determine the temperature of the water at state 2, Tz in degrees K e) (5) determine how much heat must be added to the tank to obtain state 2 f) (5) the heating continues untill the water in the tank reaches state 3 with a pressure p3-5bar. What is the temperature of the water in the tank at state 3, T3? TABLE A-3 Pressure Conversions Properties of Saturated Water (Liquid-Vapor): Pressure Table bar MP Specific Volume Internal Energy Enthalpy Entropy k/kg K Sat. Sat. Sat. Sat Sat Press Temp. Liquid Vapor Liquid Vapor bar r x10 5x 0.04 28.96 10040 34.800 0.06 36.16 10064 0.08 41.51 10084 0.10 45.81 23.739 18.103 10102 14.674 121.45 2415.2 151.53 2425.0 173.87 2432.2 191.82 2437.9 0.20 60.06 10172 7.649 0.30 69.10 10223 5.229 0.40 75.87 10265 3.993 0.50 81.33 10300 3.240 0.60 85.94 10331 2732 0.70 89.95 10360 2.365 251.38 2456.7 289.20 2468.4 317.53 2477.0 340.44 2483.9 359.79 2489.6 376.63 2494.5 Liquid Evap h hie 12146 2432.9 2554.4 0.4226 15153 2415.9 2567.4 0.5210 173.88 2403.1 2577.0 0.5926 19183 2392.8 2584.7 0.6493 8.1502 25140 2358.3 2609.7 289.23 2336.1 2625.3 0.9439 Sat. Vapor Sat Liquid Vapor Sat. Press bar 8.4746 0.04 8.3304 8.2287 0.06 0.08 0.10 0.8320 7.9085 0.20 7.7686 0.30 317.58 2319.2 2636.8 1.0259 340.49 2305.4 2645.9 1.0910 7.5939 0.50 359.86 2293.6 2653.5 1.1453 7.5320 0.60 376.70 2283.3 2660.0 1.1919 7.4797 0.70 7.6700 0.40 0.80 93.50 10380 2.087 391.58 0.90 96.71 10410 1869 405.06 2498.8 2502.6 1.00 1.50 99.63 10432 1.694 417.36 2506.1 111.4 10528 1.159 2.00 120.2 10605 0.8857 466.94 2519.7 504.49 2529.5 39166 2274.1 2665.8 1.2329 7.4346 405.15 2265.7 2670.9 1.2695 7.3949 417.46 2258.0 2675.5 1.3026 7.3594 467.11 22265 2693.6 1.4336 7.2233 504.70 22019 2706.7 1.5301 7.1271 0.80 0.90 1,00 1.50 2.00 2.50 127.4 10672 0.7187 3.00 133.6 10732 0.6058 3.50 138.9 10786 0.5243 535.10 2537.2 535.37 21815 561.15 2543.6 56147 2163.8 583.95 2546.9 584.33 2148.1 2716.9 2725.3 1.6072 7.0527 2.50 1.6718 6.9919 3.00 2732.4 1.7275 6.9405 3.50 4.00 143.6 10836 0.4625 604.31 4.50 147.9 10882 0.4140 2553.6 604.76 2133.8 622.25 2557.6 623.25 2120.7 2738.6 1.7766 6.8959 4.00 2743.9 1.8207 6.8565 4.50 5.00 151.9 10926 0.3749 639.68 2561.2 6.00 158.9 11006 0.3157 669.90 2567.4 7.00 165.0 11080 0.2729 8.00 170.4 11148 0.2404 9.00 175.4 11212 0.2150 696.44 2572.5 720.22 2576.8 741.83 2580.5 640.23 2108.5 1.8607 670.56 2086.3 2756.8 1.9312 697.22 2066.3 2763.5 1.9922 721.11 2048.0 2769.1 742.83 20311 2773.9 2748.7 6.8212 5.00 6.7600 6.00 6.7080 7.00 2.0462 6.6628 8.00 2.0946 6.6226 9.00 TABLE A-4 Properties of Superheated Water Vapor m/kg h kJ/kg kJ/kg-K m/kg p=5.0 bar=0.50 MPa (T=151.86C) kJ/kg kl/kg p=7.0 bar = 0.70 MPa (T=164.97C) kl/kg - K Sat. 0.3749 2561.2 2748.7 6.8213 180 0.4045 2609.7 2812.0 6.9656 200 0.4249 2642.9 2855.4 7.0592 0.2729 2572.5 2763.5 6.7080 320 240 0.4646 2707.6 2939.9 280 0.5034 2771.2 3022.9 0.5416 2834.7 3105.6 7.2307 7.3865 7.5308 0.2847 2599.8 2799.1 0.2999 2634.8 2844.8 0.3292 2701.8 2932.2 7.0641 0.3574 2766.9 3017.1 7.2233 0.3852 2831.3 3100.9 7.3697 6.7880 6.8865 360 0.5796 2898.7 3188.4 7.6660 440 400 0.6173 2963.2 3271.9 7.7938 0.6548 3028.6 3356.0 7.9152 0.4126 2895.8 3184.7 7.5063 0.4397 2960.9 3268.7 7.6350 0.4667 3026.6 3353.3 7.7571 500 0.7109 3128.4 3483.9 8.0873 600 0.8041 3299.6 3701.7 8.3522 700 0.8969 3477.5 3925.9 8.5952 0.5070 3126.8 3481.7 7.9299 0.5738 3298.5 3700.2 8.1956 0.6403 3476.6 3924.8 8.4391 HO P02. (30 pts) A two phase liquid-vapor mixture of HO, initially at equilibrium in state 1 with p = 1.5bar (150 kPa) with a quality of x = 75% is contained in a sealed tank with rigid walls. The mass of HO is m = 5 kg. a) (5) Determine the volume of the tank, V, in m. b) (5) Determine the total internal energy of the liquid-vapor mixture at state 1 Next, heat is added to the tank until the phase of the water in the tank becomes state 2, the saturated vapor state. c) (5) determine the pressure of the state 2 d) (5) Determine the temperature of the water at state 2, Tz in degrees K e) (5) determine how much heat must be added to the tank to obtain state 2 f) (5) the heating continues untill the water in the tank reaches state 3 with a pressure p3-5bar. What is the temperature of the water in the tank at state 3, T3? TABLE A-3 Pressure Conversions Properties of Saturated Water (Liquid-Vapor): Pressure Table bar MP Specific Volume Internal Energy Enthalpy Entropy k/kg K Sat. Sat. Sat. Sat Sat Press Temp. Liquid Vapor Liquid Vapor bar r x10 5x 0.04 28.96 10040 34.800 0.06 36.16 10064 0.08 41.51 10084 0.10 45.81 23.739 18.103 10102 14.674 121.45 2415.2 151.53 2425.0 173.87 2432.2 191.82 2437.9 0.20 60.06 10172 7.649 0.30 69.10 10223 5.229 0.40 75.87 10265 3.993 0.50 81.33 10300 3.240 0.60 85.94 10331 2732 0.70 89.95 10360 2.365 251.38 2456.7 289.20 2468.4 317.53 2477.0 340.44 2483.9 359.79 2489.6 376.63 2494.5 Liquid Evap h hie 12146 2432.9 2554.4 0.4226 15153 2415.9 2567.4 0.5210 173.88 2403.1 2577.0 0.5926 19183 2392.8 2584.7 0.6493 8.1502 25140 2358.3 2609.7 289.23 2336.1 2625.3 0.9439 Sat. Vapor Sat Liquid Vapor Sat. Press bar 8.4746 0.04 8.3304 8.2287 0.06 0.08 0.10 0.8320 7.9085 0.20 7.7686 0.30 317.58 2319.2 2636.8 1.0259 340.49 2305.4 2645.9 1.0910 7.5939 0.50 359.86 2293.6 2653.5 1.1453 7.5320 0.60 376.70 2283.3 2660.0 1.1919 7.4797 0.70 7.6700 0.40 0.80 93.50 10380 2.087 391.58 0.90 96.71 10410 1869 405.06 2498.8 2502.6 1.00 1.50 99.63 10432 1.694 417.36 2506.1 111.4 10528 1.159 2.00 120.2 10605 0.8857 466.94 2519.7 504.49 2529.5 39166 2274.1 2665.8 1.2329 7.4346 405.15 2265.7 2670.9 1.2695 7.3949 417.46 2258.0 2675.5 1.3026 7.3594 467.11 22265 2693.6 1.4336 7.2233 504.70 22019 2706.7 1.5301 7.1271 0.80 0.90 1,00 1.50 2.00 2.50 127.4 10672 0.7187 3.00 133.6 10732 0.6058 3.50 138.9 10786 0.5243 535.10 2537.2 535.37 21815 561.15 2543.6 56147 2163.8 583.95 2546.9 584.33 2148.1 2716.9 2725.3 1.6072 7.0527 2.50 1.6718 6.9919 3.00 2732.4 1.7275 6.9405 3.50 4.00 143.6 10836 0.4625 604.31 4.50 147.9 10882 0.4140 2553.6 604.76 2133.8 622.25 2557.6 623.25 2120.7 2738.6 1.7766 6.8959 4.00 2743.9 1.8207 6.8565 4.50 5.00 151.9 10926 0.3749 639.68 2561.2 6.00 158.9 11006 0.3157 669.90 2567.4 7.00 165.0 11080 0.2729 8.00 170.4 11148 0.2404 9.00 175.4 11212 0.2150 696.44 2572.5 720.22 2576.8 741.83 2580.5 640.23 2108.5 1.8607 670.56 2086.3 2756.8 1.9312 697.22 2066.3 2763.5 1.9922 721.11 2048.0 2769.1 742.83 20311 2773.9 2748.7 6.8212 5.00 6.7600 6.00 6.7080 7.00 2.0462 6.6628 8.00 2.0946 6.6226 9.00 TABLE A-4 Properties of Superheated Water Vapor m/kg h kJ/kg kJ/kg-K m/kg p=5.0 bar=0.50 MPa (T=151.86C) kJ/kg kl/kg p=7.0 bar = 0.70 MPa (T=164.97C) kl/kg - K Sat. 0.3749 2561.2 2748.7 6.8213 180 0.4045 2609.7 2812.0 6.9656 200 0.4249 2642.9 2855.4 7.0592 0.2729 2572.5 2763.5 6.7080 320 240 0.4646 2707.6 2939.9 280 0.5034 2771.2 3022.9 0.5416 2834.7 3105.6 7.2307 7.3865 7.5308 0.2847 2599.8 2799.1 0.2999 2634.8 2844.8 0.3292 2701.8 2932.2 7.0641 0.3574 2766.9 3017.1 7.2233 0.3852 2831.3 3100.9 7.3697 6.7880 6.8865 360 0.5796 2898.7 3188.4 7.6660 440 400 0.6173 2963.2 3271.9 7.7938 0.6548 3028.6 3356.0 7.9152 0.4126 2895.8 3184.7 7.5063 0.4397 2960.9 3268.7 7.6350 0.4667 3026.6 3353.3 7.7571 500 0.7109 3128.4 3483.9 8.0873 600 0.8041 3299.6 3701.7 8.3522 700 0.8969 3477.5 3925.9 8.5952 0.5070 3126.8 3481.7 7.9299 0.5738 3298.5 3700.2 8.1956 0.6403 3476.6 3924.8 8.4391 HO
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
A pipe carries steam as a two phase liquid vapor mixture at 2.0 MPa. A small quantity is withdrawn through a throttling calorimeter, where it undergoes a throttling process to an exit pressure of 0.1...
-
Furniture Co. incurred the following costs during 2016: Conversion costs Prime costs Manufacturing overhead What was the amount of direct materials and direct labor used for the year? Direct...
-
A hollow conducting sphere of radius R carries a negative charge q. (a) Write expressions for the electric field E inside (r < R) and outside (r > R) the sphere. Also indicate the direction of the...
-
Choose a set S of three points such that aff S is the plane in R 3 whose equation is x 3 = 5. Justify your work.
-
Mrs. Clarks Foods was an Iowa company engaged in the business of distributing juice beverages. International Suntrade and Miller & Smith Foods were Canadian companies that acted as brokers...
-
Use the following information to prepare a classified balance sheet for Alpha Co. at the end of 2016. Accounts receivable .....$26,500 Accounts payable ....... 12,200 Cash ............ 20,500 Common...
-
2 Charlie's Crispy Chicken (CCC) operates a fast-food restaurant. When accounting for its first year of business, CCC created several accounts. Account Name Description Balance Accounts Payable...
-
1. Rafter Company had the following accounts and balances at December 31, 2018: Equipment Cash A A 205,000 Unearned Revenue 12,000 Inventory 38,000 Salary Expense 65,000 Note Payable - 1 year Common...
-
The author's final conclusion is that "increased concentration in food retailing (due to increasing consolidation and foreign investment) has resulted in food prices increases, as retailers get their...
-
Question 1 A basic proposition is that exchange rates adjust so as to maintain the purchasing power parity (PPP): the price of a bundle of goods, expressed in common currency, should be the same...
-
A computer system is required that will support the following small garage business : Customers bring their cars to the garage for servicing and repair. The attendant must check the car in, record...
-
How does the process of endosymbiosis contribute to the evolution of eukaryotic cells, and what evidence supports this theory?
-
find an example problem that can be solved using Machine Learning. In this regards, show how does Machine Learning approach solve that problem. Please thoroughly describes ?!
-
John Deere CEO Samuel Allen aims to increase total sales to $50 billion by 2018, with half coming from outside the U.S. and Canada, up from 39 percent today. In the last few years, the company has...
-
What are the main distinctions between the different schools of legal interpretation?
-
Let \(X, Y, X_{n}, Y_{n}: \Omega ightarrow \mathbb{R}, n \geqslant 1\), be random variables. a) If, for all n > 1, Xn Yn and if (Xn, Yn) (X, Y), then XIL Y. b) Let X Y such that X, Y ~ B1/2 = (80...
-
Let \(X_{n}, Y_{n}: \Omega ightarrow \mathbb{R}, n \geqslant 1\), be two sequences of random variables. a) If \(X_{n} \xrightarrow{d} X\) and \(Y_{n} \xrightarrow{\mathbb{P}} c\), then \(X_{n} Y_{n}...
-
Let \(X_{n}, Y_{n}: \Omega ightarrow \mathbb{R}^{d}, n \geqslant 1\), be two sequences of random variables such that \(X_{n} \xrightarrow{d} X\) and \(X_{n}-Y_{n} \xrightarrow{\mathbb{P}} 0\). Then...

Study smarter with the SolutionInn App


