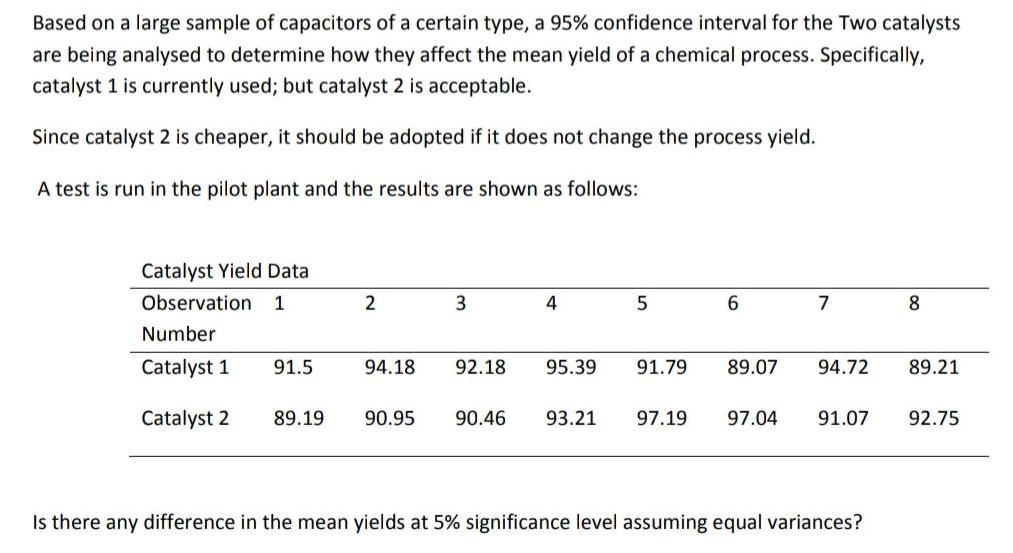
Based on a large sample of capacitors of a certain type, a 95% confidence interval for...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Based on a large sample of capacitors of a certain type, a 95% confidence interval for the Two catalysts are being analysed to determine how they affect the mean yield of a chemical process. Specifically, catalyst 1 is currently used; but catalyst 2 is acceptable. Since catalyst 2 is cheaper, it should be adopted if it does not change the process yield. A test is run in the pilot plant and the results are shown as follows: Catalyst Yield Data Observation 1 2 3. 4 7 8. Number Catalyst 1 91.5 94.18 92.18 95.39 91.79 89.07 94.72 89.21 Catalyst 2 89.19 90.95 90.46 93.21 97.19 97.04 91.07 92.75 Is there any difference in the mean yields at 5% significance level assuming equal variances? Based on a large sample of capacitors of a certain type, a 95% confidence interval for the Two catalysts are being analysed to determine how they affect the mean yield of a chemical process. Specifically, catalyst 1 is currently used; but catalyst 2 is acceptable. Since catalyst 2 is cheaper, it should be adopted if it does not change the process yield. A test is run in the pilot plant and the results are shown as follows: Catalyst Yield Data Observation 1 2 3. 4 7 8. Number Catalyst 1 91.5 94.18 92.18 95.39 91.79 89.07 94.72 89.21 Catalyst 2 89.19 90.95 90.46 93.21 97.19 97.04 91.07 92.75 Is there any difference in the mean yields at 5% significance level assuming equal variances?
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
Based on a large sample of capacitors of a certain type, a 95% confidence interval for the mean capacitance, in F, was computed to be (0.213, 0.241). Find a 90% confidence interval for the mean...
-
A student wanted to construct a 95% confidence interval for the mean age of students in her statistics class She randomly selected nine students Their average age was 19.1 years with a sample...
-
Assuming normality, find a 95% confidence interval for the variance from the sample 145.3, 145.4, 146.2.
-
As manager of a local pizza parlor, you want to develop a balanced scorecard so you can more effectively monitor the restaurants performance. Required a. Propose at least two goals for each...
-
J. Greene and J. Touchstone conducted a study on the relationship between the estriol levels of pregnant women and the birth weights of their children. Their findings, "Urinary Tract Estriol: An...
-
Figure shows the tensile engineering stress?strain behavior for a steel alloy. (a) What is the modulus of elasticity? (b) What is the proportional limit? (c) What is the yield strength at a strain...
-
Environmental Concerns Limited (ECL) issued \$500,000 of 10-year, 6\% bonds payable at maturity (par) value on May 1, 2008. The bonds pay interest each April 30 and October 31, and the company ends...
-
1. Discuss and contrast the features of the retirement plans offered by Analytical Solutions and Precision Manufacturing. 2. Which plan do you think is more desirable? Consider the features,...
-
2. Delta Corp. has $12,600 in accounts receivable; $64,800 in inventory; cash of $5,150; notes payable: $6900; Net fixed assets: $278,650; accounts payable: $4,800; Long-term debt: $152,700....
-
Three-Month Project NOTE! Templates needed Ampersand, Inc., is a small business that operates in Somerset, VT The company is located at 732 Appalachian Way, Somerset, VT 05363. Its federal Employer...
-
Consider the formula below for determining the percent of capital gain earned on an investment. p = 100 (y-w/w) which of the following equations is a correct equivalent equation solved for a...
-
Based on your personal experience as a consumer and your marketing knowledge, what would you do differently from at least one of the companies reviewed and why?
-
Summarize the talent management process. What is the difference between Job Description and Job Specification? How can workflow analysis be used to create a job description? What is the difference...
-
In order to protect domestic jobs, country Alpha places a tariff on imported goods from country Bravo. a) What happens to the price of Alpha's imported goods from Bravo? What happens to the quantity...
-
How does self-interest impact the marketing process? How could marketers convince an audience to support funding for after-school program services that they do not use? Provide an example of an...
-
Bims Corporation uses the weighted-average method in its process costing system. The Assembly Department started the month with 6,000 units in its beginning work in process inventory that were 90%...
-
Your client was fired from her job. She claims the employer engaged in discrimination and that the employer's actions violated the law. Your client worked for the employer in Maryland. The employer...
-
Imagine that your best friend knows you are taking a psychology course and wonders what psychology is all about. How would you define psychology for your friend? Write an essay on the discipline of...
-
Bolts manufactured for a certain purpose may be classified as acceptable (suitable for the intended purpose), downgraded (unsuitable for the intended purpose but acceptable for a different purpose),...
-
A mobile computer network consists of a number of computers (called nodes) that communicate with each other while moving throughout a region. A node that is out of transmission range of the other...
-
Refer to Exercise 5. a. Assume g = 9.80 m/s2 exactly, and that L = 0.855 0.005 m. Estimate T, and find the relative uncertainty in the estimate. b. Assume L = 0.855 m exactly, and that T = 1.856 ...
-
(a) Describe the effect of temperature on the equilibrium constant. (b) Industrial \(\mathrm{CH}_{3} \mathrm{OH}\) is prepared according to the reaction \[ \mathrm{CO}(\mathrm{g})+2...
-
Mention the areas of application of the fuel cell.
-
Estimate the standard free energy change and equilibrium constant at \(700 \mathrm{~K}\) for the reaction \[ \mathrm{N}_{2}(\mathrm{~g})+3 \mathrm{H}_{2}(\mathrm{~g})=2 \mathrm{NH}_{3}(\mathrm{~g})...

Study smarter with the SolutionInn App


