QUESTION 5 Which of the following are good leaving groups? Choose all that apply. a. -OH...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
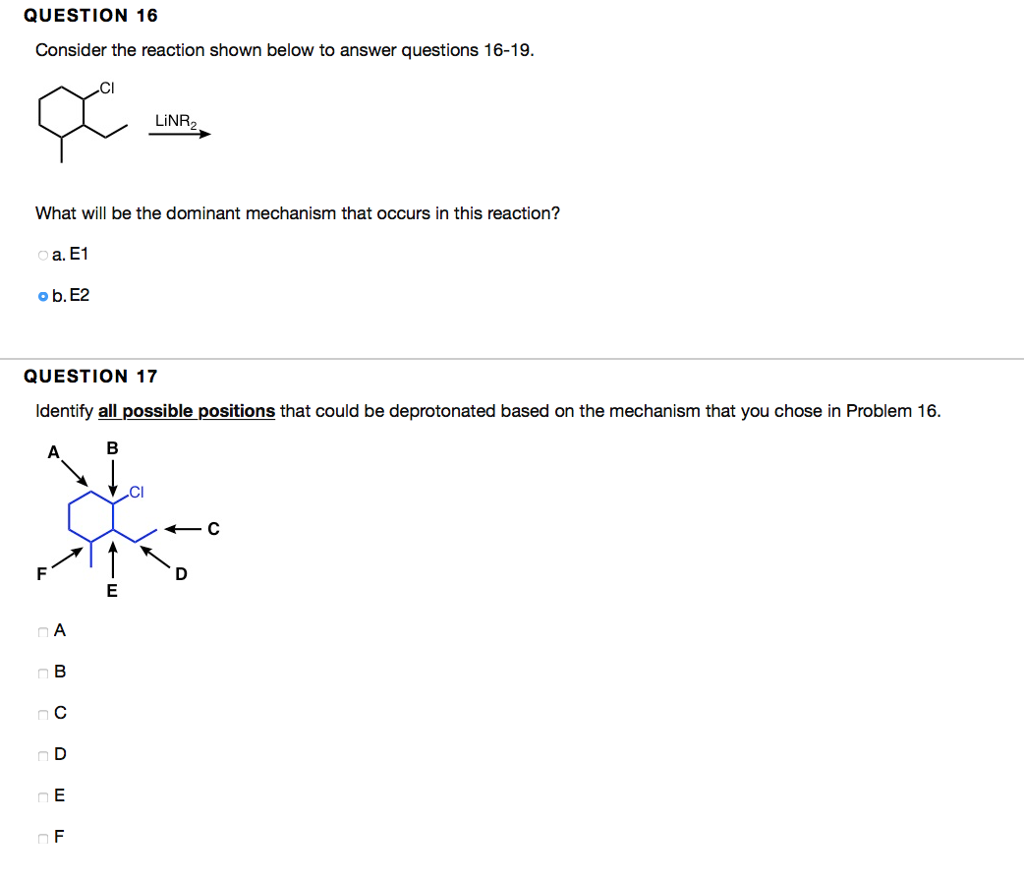
QUESTION 5 Which of the following are good leaving groups? Choose all that apply. a. -OH b.-OTf c. -Br d.-OMs e. -Cl 1. -OTS g.-F HO QUESTION 6 1 points Save Answer For problems 6-14, complete the reactions by filling the boxes with the necessary chemical reagent(s)/reaction conditions or major products. Provide reagent(s)/reaction conditions that would produce the major product most efficiently. Assume aqueous workups are performed if needed. OH a. Ag2O, NH4OH, H₂O b. NaBH4 c. CrO3, HCl, pyridine d. NaOH e, HBr f. H₂SO4 g. CrO3, H₂SO4, H₂O h. LIAIH4 oi. H₂O 0.25 points OH Save Answer QUESTION 16 Consider the reaction shown below to answer questions 16-19. x What will be the dominant mechanism that occurs in this reaction? oa. E1 ob. E2 QUESTION 17 Identify all possible positions that could be deprotonated based on the mechanism that you chose in Problem 16. B A A B C D E F A о в nc OD DE LINR₂ OF E ← C D Consider the dehydration reaction using the starting material shown below to answer questions 20-23. Hoft HO Which reagent(s) are required to perform a dehydration reaction? oa. HCI ob. H2SO4 oc. H₂O od. NaOH o e. H₂SO4, heat of. NaOH, heat Og. NaH QUESTION 21 Identify all carbocations that appear in the mechansim for this reaction. Choose all that apply. i) na.i b.ii nc. iii Dehydration nd.iv ii) iii) iv) e. None of the above. The reaction goes by an E2 mechanism and no carbocations are formed. QUESTION 5 Which of the following are good leaving groups? Choose all that apply. a. -OH b.-OTf c. -Br d.-OMs e. -Cl 1. -OTS g.-F HO QUESTION 6 1 points Save Answer For problems 6-14, complete the reactions by filling the boxes with the necessary chemical reagent(s)/reaction conditions or major products. Provide reagent(s)/reaction conditions that would produce the major product most efficiently. Assume aqueous workups are performed if needed. OH a. Ag2O, NH4OH, H₂O b. NaBH4 c. CrO3, HCl, pyridine d. NaOH e, HBr f. H₂SO4 g. CrO3, H₂SO4, H₂O h. LIAIH4 oi. H₂O 0.25 points OH Save Answer QUESTION 16 Consider the reaction shown below to answer questions 16-19. x What will be the dominant mechanism that occurs in this reaction? oa. E1 ob. E2 QUESTION 17 Identify all possible positions that could be deprotonated based on the mechanism that you chose in Problem 16. B A A B C D E F A о в nc OD DE LINR₂ OF E ← C D Consider the dehydration reaction using the starting material shown below to answer questions 20-23. Hoft HO Which reagent(s) are required to perform a dehydration reaction? oa. HCI ob. H2SO4 oc. H₂O od. NaOH o e. H₂SO4, heat of. NaOH, heat Og. NaH QUESTION 21 Identify all carbocations that appear in the mechansim for this reaction. Choose all that apply. i) na.i b.ii nc. iii Dehydration nd.iv ii) iii) iv) e. None of the above. The reaction goes by an E2 mechanism and no carbocations are formed.
Expert Answer:
Answer rating: 100% (QA)
5 Ans a OH This is a poor leaving group because the hydroxide ion is a strong base and prefers to retain its electron pair b OTf tosylate This is a good leaving group because it is stable and can leav... View the full answer

Related Book For 

Fundamentals of biochemistry Life at the Molecular Level
ISBN: 978-0470547847
4th edition
Authors: Donald Voet, Judith G. Voet, Charlotte W. Pratt
Posted Date:
Students also viewed these chemistry questions
-
Identify with a which of the following are subdivisions of owners equity. a. Vehicles _____ b. J. Penny, Capital _____ c. Accounts Payable _____ d. J. Penny, Withdrawals _____ e. Accounts Receivable...
-
Identify with a which of the following are subdivisions of owners equity. a. Land b. M. Kaminsky, Capital c. Accounts Receivable d. M. Kaminsky, Withdrawals _____ e. Accounts Payable _____ f. Rent...
-
Which of the following are Section 1231 assets? Explain. Assume all the items have been held long-term. a. Machinery used in the business b. Personal home c. Factory building d. Land held as an...
-
Your buddy mentioned that she is thinking about suing under the qui tam law. You are not sure what that is. a. What is a qui tam provision? b. Can employees who do not work for the government sue...
-
Lynn transfers land having a $50,000 adjusted basis, an $80,000 FMV, and $10,000 cash to Allied Corporation in exchange for 100% of allied stock. The corporation assumes the $70,000 mortgage on the...
-
The maximum time to complete a task in a project is 2.5 days. Suppose that the completion time as a proportion of this maximum is a beta random variable with = 2 and = 3. What is the probability...
-
Mid America Amusements Corporation bad the following stockholders' equity on November 30: On December 30, Mid America purchased 10,000 shares of treasury stock at \(\$ 9\) per share. 1. Journalize...
-
Into what two categories does the FASAB divide government assets? How are each of the two accounted for?
-
The partnership of Waterway, Wildhorse, and Sheffield engaged you to adjust its accounting records and convert them uniformly to the accrual basis in anticipation of admitting Kerns as a new partner....
-
Zek Ltd assembles heavy industrial switchboxes. Hitherto, Zek Ltd has assembled only one type, but recently a decision was made to expand the product range into two types. The following data pertains...
-
An engineering summer student is asked to determine the surface tension of a sample of liquid. She has access to thin wire of various diameters and conducts a series of experiments. The wire is made...
-
1. What is trencher bread? 2. What does a 'Mille-Feuille' look like? 3. Research and provide baking temperature and cooking times for the following (you should reference your source for assessor...
-
A 16-gram mass is moving to the right at 3 x 10^-1 m/s while a 4.0 x 10-3-kilogram mass is moving to the left at 50-centimeters/second. They collide head on and stick together. Find their velocity...
-
1. A toy car of mass 1.2 kg is driving vertical circles inside a hollow cylinder of radius 2.0m. It is moving at a constant speed of 6 m/s. a) Calculate the magnitude of the normal force acting on...
-
How does McDonalds handle its expatriate managers? Does McDonalds have issues or challenges related to expatriates?
-
find an article about a business financial failure (with a good ending or a bad one). PLEASE LINK THE ARTICLE 1. Provide the name of the business, the owner(s), and what form of business ownership it...
-
The column shown in the figure is fixed at the base and free at the upper end. A compressive load P acts at the top of the column with an eccentricity e from the axis of the column. Beginning with...
-
Assume that k2 = kcat for a highly purified enzyme for which you seek to determine G. Using the Arrhenius equation, k = Ae-G/RT, where k is a reaction rate constant and A is a constant, you can...
-
Explain how the following affect the Tm of double-stranded DNA: (a) Increasing the monovalent salt concetration. (b) Decreasing the pH. (c) Increasing the pH. (d) Increasing the concentration of...
-
The leaves of some species of desert plants taste sour in the early morning, but, as the day wears on, they become tasteless and then bitter. Explain.
-
For the data set a. Find the first and third quartiles. b. Find the IQR. c. Find the upper and lower outlier boundaries. d. List all the values, if any, that are classified as outliers. 15 7 2 4 4 3...
-
A population has mean = 25 and standard deviation = 4. a. Find the z-score for a population value of 16. b. Find the z-score for a population value of 31. c. What number has a z-score of 2.5?
-
For the data set 37 82 20 25 31 10 41 44 4 36 68 a. Find the first and third quartiles. b. Find the IQR. c. Find the upper and lower outlier boundaries. d. List all the values, if any, that are...

Study smarter with the SolutionInn App


