Question: (a) Determine the number and activities of the carbonyl stretching modes of both the cis and trans isomers of L2M(CO)4. (5 marks) (b) Calculate
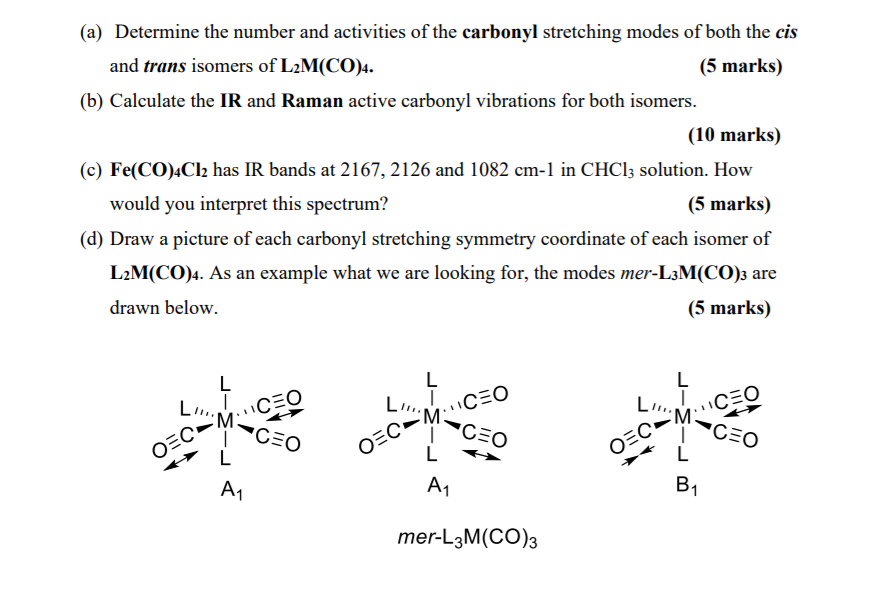
(a) Determine the number and activities of the carbonyl stretching modes of both the cis and trans isomers of L2M(CO)4. (5 marks) (b) Calculate the IR and Raman active carbonyl vibrations for both isomers. (10 marks) (c) Fe(CO)4Cl2 has IR bands at 2167, 2126 and 1082 cm-1 in CHCl3 solution. How would you interpret this spectrum? (5 marks) (d) Draw a picture of each carbonyl stretching symmetry coordinate of each isomer of L2M(CO)4. As an example what we are looking for, the modes mer-L3M(CO)3 are drawn below. (5 marks) LI. OEC L M A CEO CEO LI.. O=C M CEO CEO L A mer-L3M(CO) 3 OEC- M: L B CEO CEO
Step by Step Solution
3.51 Rating (154 Votes )
There are 3 Steps involved in it
Per 1 co C2 3 Co NUSA 3 21 ... View full answer

Get step-by-step solutions from verified subject matter experts


