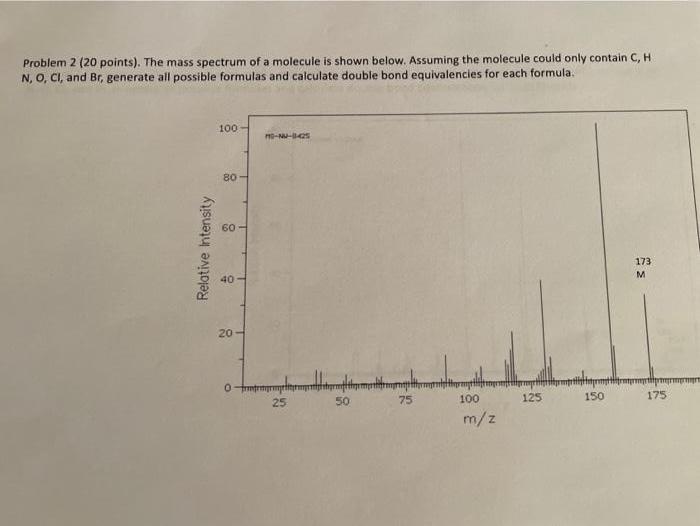
Problem 2 (20 points). The mass spectrum of a molecule is shown below. Assuming the molecule...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Problem 2 (20 points). The mass spectrum of a molecule is shown below. Assuming the molecule could only contain C, H N, O, CI, and Br, generate all possible formulas and calculate double bond equivalencies for each formula. 100 80- 60 173 40 20- 0mtrmrmt 25 50 75 100 125 150 175 m/z Relative Intensity Problem 2 (20 points). The mass spectrum of a molecule is shown below. Assuming the molecule could only contain C, H N, O, CI, and Br, generate all possible formulas and calculate double bond equivalencies for each formula. 100 80- 60 173 40 20- 0mtrmrmt 25 50 75 100 125 150 175 m/z Relative Intensity
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The molecular ion region in the mass spectrum of a large molecule, such as a protein, consists of a cluster of peaks differing by 1 Da. This pattern occurs because a molecule with many atoms has a...
-
The mass spectrum of methyl isobutyl ether does not show a peak due to inductive cleavage, in contrast to the mass spectrum of di-sec-butyl ether (Eq. 12.31). Use what you know about carbocation...
-
The mass spectrum of 1-butanol shows an intense daughter ion peak at m/z 5 31. Explain how this peak might arise.
-
Write a program for setting three bits of an unsigned char to 1. The function set 3bits should have four parameters: the unsigned char to be changed and the three bits which are to be set to 1. For...
-
Systems and Software, Inc. ( SAS), located in Colchester, Vermont, designs, develops, sells, and services software that allows utility providers to organize their data, including customer...
-
Beeler Furniture Company deposits all cash receipts each Wednesday and Friday in a night depository, after banking hours. The data required to reconcile the bank statement as of June 30, 20Y2, have...
-
People have proposed driving motors with the earth's magnetic field. This is possible in principle, but the small field means that unrealistically large currents are needed to produce noticeable...
-
The adjusted trial balance for Tybalt Construction as of December 31, 2017, follows. O. Tybalt invested $5,000 cash in the business during year 2017 (the December 31, 2016, credit balance of the O....
-
In initial public offerings (IPOs) relatively few firms pay cash dividends. Why do you suppose that is? Explain.
-
What is a balanced scorecard? What is its primary objective?
-
Product Customization - android and IOS platforms Innovation Price Comparison pricing- Alibaba subscription Promotion Social media - Chevrolet influencers. a- Factors affecting marketing mix due to...
-
Project: Implementing a patient portal in rehabilitation and specialty hospitals Explanatory details needed Evaluate what type of project information needs to be communicated to stakeholders...
-
Throughout the course, they created deliverables that were incorporated into an adaptive leadership toolkit submitted in Module Seven. They will recall this toolkit should help improve the skills and...
-
Power and politics tend to have negative connotations. Present arguments for and against this notion in the context of an organization. Cite relevant examples to support your views.
-
Every person has a certain degree of emotional intelligence (EQ); we may not be fully aware of when and how we use it. EQ helps us to build stronger relationships, succeed professionally and...
-
You are valuing a four year project. The initial capital expenditure (capex) is $125 million. You have a risk free agreement to sell the PP&E four years from now for $80 million. The relevant...
-
What is meant by the term "recession"? How fast did Real GDP grow in the 4 th Quarter of 2022 and 2 nd Quarter of 2023? Will the FED's monetary policy result in a recession in the U.S. economy in...
-
Write an SQL statement to display all data on products having a QuantityOnHand greater than 0.
-
Classify the following peptides as acidic, basic, or neutral. What is the net charge on each peptide at pH = 6? (a) Gly-Leu-Val (b) Leu-Trp-Lys-Gly-Lys (c) N-acetyl-Asp-Val-Ser-Arg-Arg (A-acetyl...
-
From the molecular masses and the relative intensities of their M and M + 1 peaks, suggest molecular formulas for the following compounds. M (m/z = 82; 37%), M + 1 (2.5%); contains C and H.
-
Predict the predominant products that would result from bromination of each of the following compounds, classify each substituent group as an ortho, para director or a meta director, and explain your...
-
What is a program?
-
In a(n)________ numbering system, all numeric values are written as sequences of 0s and 1s. a. hexadecimal b. binary c. octal d. decimal
-
What encoding scheme is extensive to represent all the characters of all the languages in the world?

Study smarter with the SolutionInn App



