Question: You have a stock solution containing 1.25 mM of substrate in borate buffer pH 9.5. You transfer 1.25 mL of this stock into a
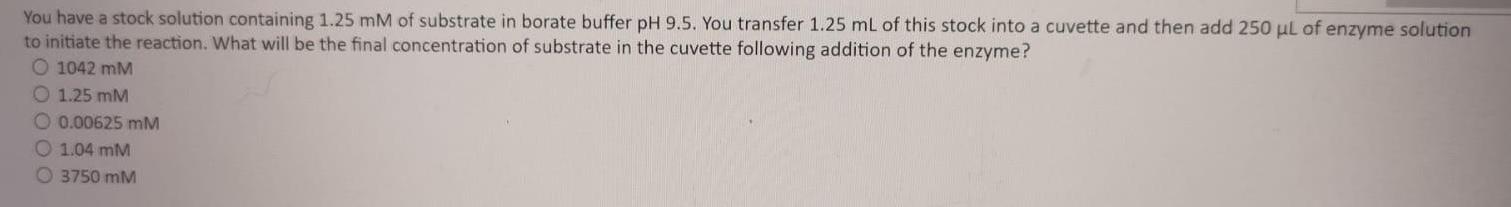
You have a stock solution containing 1.25 mM of substrate in borate buffer pH 9.5. You transfer 1.25 mL of this stock into a cuvette and then add 250 ul of enzyme solution to initiate the reaction. What will be the final concentration of substrate in the cuvette following addition of the enzyme? O 1042 mM O 1.25 mM O 0.00625 mM O 1.04 mM O 3750 mM
Step by Step Solution
3.50 Rating (153 Votes )
There are 3 Steps involved in it
Ans 104 mM Explanation Taking125 ml of the stock so... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (2 attachments)
60d02c454241a_219570.pdf
180 KBs PDF File
60d02c454241a_219570.docx
120 KBs Word File


