Question: Solid magnesium selenide has a crystal structure with the cubic unit cell pictured below, where the Se ions are represented as green spheres. The
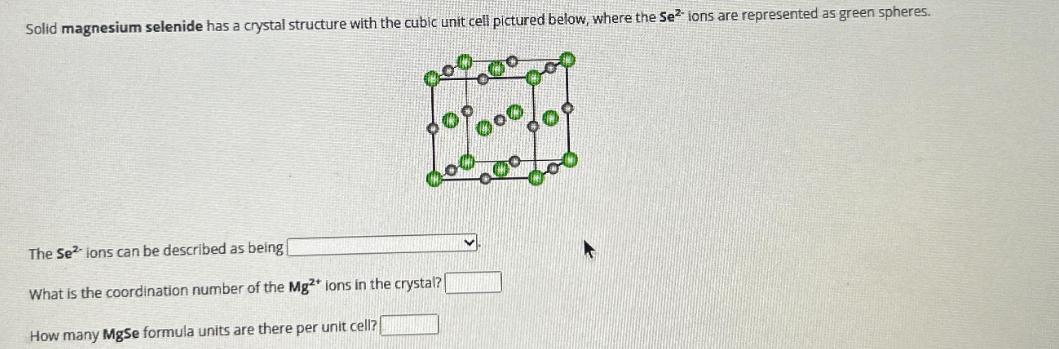
Solid magnesium selenide has a crystal structure with the cubic unit cell pictured below, where the Se ions are represented as green spheres. The Se ions can be described as being What is the coordination number of the Mg2+ ions in the crystal? How many MgSe formula units are there per unit cell?
Step by Step Solution
There are 3 Steps involved in it
Solutions SOLUTION 2 forms ccp since it is large ion occupyi... View full answer

Get step-by-step solutions from verified subject matter experts


