The oxidation numbers of P, S, and Cl in HPO, HS, and KC1O4 are, respectively A)...
Fantastic news! We've Found the answer you've been seeking!
Question:
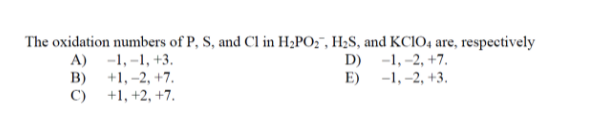
Transcribed Image Text:
The oxidation numbers of P, S, and Cl in H₂PO₂, H₂S, and KC1O4 are, respectively A) -1,-1, +3. D) -1, -2, +7. B) +1, -2, +7. E) -1, -2, +3. C) +1, +2, +7. The oxidation numbers of P, S, and Cl in H₂PO₂, H₂S, and KC1O4 are, respectively A) -1,-1, +3. D) -1, -2, +7. B) +1, -2, +7. E) -1, -2, +3. C) +1, +2, +7.
Expert Answer:
Answer rating: 100% (QA)
H2PO2 Through the periodic table H lies in 1st group having an oxidation state of 1 O lies ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A 4-lb collar can slide without friction along a horizontal rod and is in equilibrium at A when it is pushed 1 in. to the right and released from rest. The springs are undeformed when the collar is...
-
Q.1) The frontal rectangle ABCD in space placed in position that the point A is (2,2,3). The true length of its side AB is 5 cm and its degree of inclination to the horizontal plane is 40, and the...
-
The oxidation numbers of Cu and Bi in high-temperature superconductors of the type Bi 2 Sr 2 (Ca 0.8 Y 0. 2)Cu 2 O x (which could contain Cu 2+ , Cu 3+ , Bi 3+ , and Bi 5+ ) can be measured by the...
-
List the following numbers in numerical order, starting with the smallest: 1, 9, , 5, |8| , |3|, 3.1.
-
In December of 20x4, Memphis Plastics sold a forklift for $9,255. The machine was purchased in 20x1 for $50,000. Since then $38,845 in depreciation has been recorded on the forklift. Required: 1....
-
A vertical spring stores 0.962 J in spring potential energy when a 3.5-kg mass is suspended from it. (a) By what multiplicative factor does the spring potential energy change if the mass attached to...
-
We will look at walks on a network. Consider a network of N nodes. Assume that each pair of nodes are connected by a link with probability p. In the following we neglect loops and work in the limit...
-
Tucson Tractor Inc. has accumulated the following data over a six-month period: Separate the indirect labor into its fixed and variable components, using the high-lowmethod. Indirect Labor Hours 400...
-
Is there any resource that you could consider as a distinctive competence for Yunnan Lucky Air?
-
Niagara Dairy is a boutique cheese maker based in the Niagara region of Ontario. Over the years, the business has grown by supplying local retailers and, eventually, by exporting cheese products. In...
-
Explain the Characteristics of Ideal-Typical Bureaucracy?
-
Initial reaction is based on pure intuitive sense.* A. TRUE B. FALSE Concepts embodying new art and entertainment are easy to test.* A. TRUE B. FALSE
-
Seagate Limited is a steel manufacturing company which have a turnover of 543 Million Rupees. Recently, Mr. Ramanujan joined the Seagate Limited as a CEO. Mr. Ramanujan is a technically-educated CEO...
-
Entrepreneurs are always looking for unique opportunities to fill needs or wants.* A. TRUE B. FALSE The easiest step in the creative endeavor is the implementation and evaluation phase.* A. TRUE B....
-
Big Red Trucking is considering the acquisition of MidWest Transportation. MidWest's asking price of $ 5 3 5 million appears to be non - negotiable. Big Red plans to value MidWest's cash flows using...
-
The effect of arbitrage transactions on markets can best be described as: a. currency and money markets will be destabilized. b. those who need currencies for business and trade purposes will be...
-
If the interest rate is 6% and you are planing to sell your house to Ali. he offered down payment of 10000 and the rest of your money 50000 yearly for two years another 75000 in three years. how much...
-
A routine activity such as pumping gasoline can be related to many of the concepts studied in this text. Suppose that premium unleaded costs $3.75 per gal. Work Exercises in order. Use the...
-
A chromatographic procedure separates 4.0 mg of unknown mixture on a column with a length of 40 cm and a diameter of 0.85 cm. (a) What size column would you use to separate 100 mg of the same...
-
(a) Potassium iodate solution was prepared by dissolving 1.022 g of KIO3 (FM 214.00) in a 500-mL volumetric flask. Then 50.00 mL of the solution were pipetted into a flask and treated with excess KI...
-
(a) How many electrons are in one coulomb? (b) How many coulombs are in one mole of charge?
-
The primary problem with the hypothesis that life on Earth originated on Mars is that (a) Mars has never had water. (b) the proposed Martian fossils are much smaller than the tiniest bacteria on...
-
Which of these statements regarding the origin of life is false? (a) Life originated on an Earth whose atmosphere contained high levels of oxygen. (b)Miller and Urey obtained amino acids and other...
-
When you carry shopping bags, rather than grasp the handles with your hand as in Figure Q8.14a, you might choose to put them over your arm and slide the handle toward your elbow as in Figure Q8.14b....

Study smarter with the SolutionInn App


