You have been engaged as a consultant to the chemical company that is evaluating two contractor bids
Question:
You have been engaged as a consultant to the chemical company that is evaluating two contractor bids on a site cleanup project. As you would expect, the costs of the processes are related to the percent of impurities that will be removed from the soil. From past experience on the two processes that will be used by the two bidding companies, the chemical company has determined that an estimated cost function for the process to be used by Briggs Environmental is given by the rule cB= [5.2p/(96-p)] +0.15p and the estimated cost for the process used by Jason's Cleanup is given by the rule cJ=[5p/(95-p)] - 0.1p where p is the percent of impurities that will be removed and the cost is in THOUSANDS of dollars.
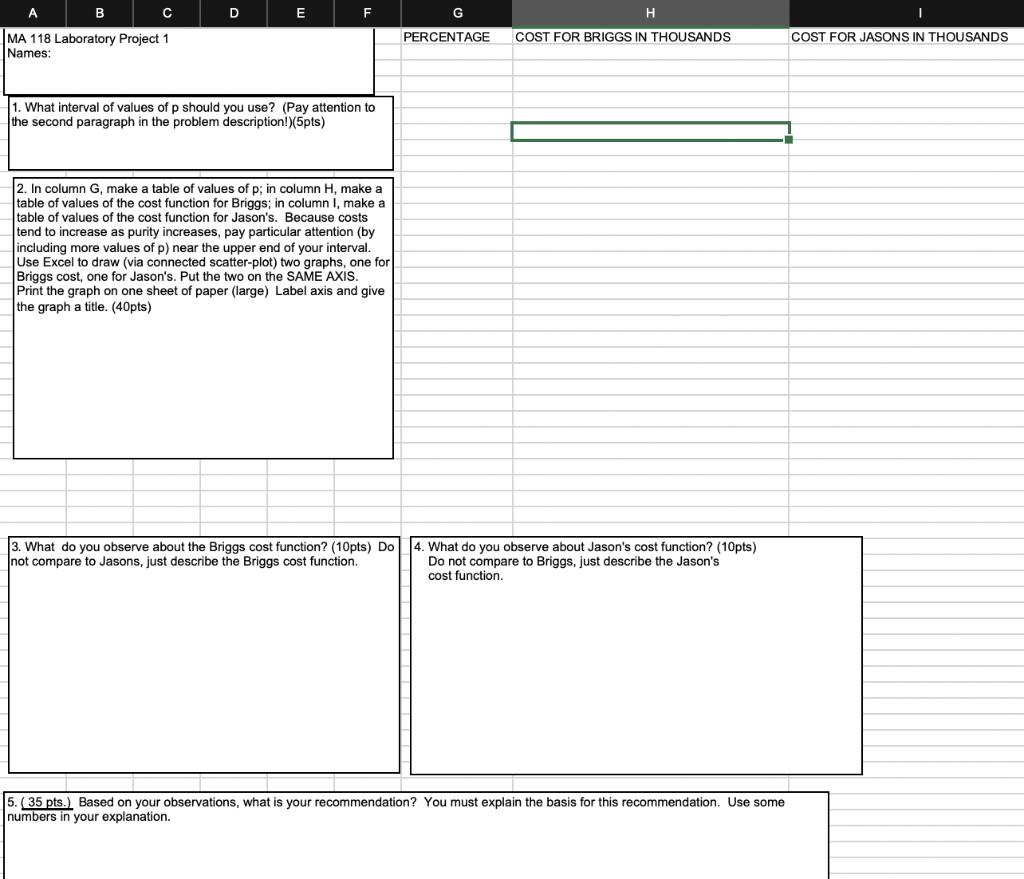 Note: since p is the percent of impurities removed, it ranges in value from 0 to 100 inclusive.The CEO of the chemical company would prefer to use the Jason's, since there has been a long standing relationship with them. The problem is that the chemical company must award the contract before the final regulations are available from the DEP on the required level of impurities that must be removed at the site. At this point, the ruling could be anywhere from 87 to 95% removal.As a consultant, you are to prepare a report analyzing the two contractors and their associated cost functions. Your report should describe a decision making method for choosing between the company Briggs and Jason's, recognizing that the CEO of the chemical company favors Jason's; yet the rules on what percent of the impurities must be removed have not as yet be established by the DEP, and the CEO will be fired if the company's costs are excessive. Your report should be written on the next sheet (entitled "Your Solution") and that is the only sheet you should hand in. Be sure, if it needs more than one page, that your names are on all pages.
Note: since p is the percent of impurities removed, it ranges in value from 0 to 100 inclusive.The CEO of the chemical company would prefer to use the Jason's, since there has been a long standing relationship with them. The problem is that the chemical company must award the contract before the final regulations are available from the DEP on the required level of impurities that must be removed at the site. At this point, the ruling could be anywhere from 87 to 95% removal.As a consultant, you are to prepare a report analyzing the two contractors and their associated cost functions. Your report should describe a decision making method for choosing between the company Briggs and Jason's, recognizing that the CEO of the chemical company favors Jason's; yet the rules on what percent of the impurities must be removed have not as yet be established by the DEP, and the CEO will be fired if the company's costs are excessive. Your report should be written on the next sheet (entitled "Your Solution") and that is the only sheet you should hand in. Be sure, if it needs more than one page, that your names are on all pages.

Smith and Roberson Business Law
ISBN: 978-0538473637
15th Edition
Authors: Richard A. Mann, Barry S. Roberts




