Explain the following reaction and the indicated stereochemical result mechanistically. H CH3 1. AICI, 2. H,
Question:
Explain the following reaction and the indicated stereochemical result mechanistically.
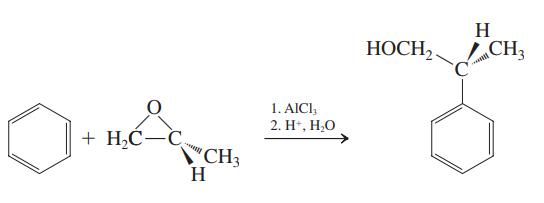
Transcribed Image Text:
H НОСН CH3 1. AICI, 2. H", Н.О + H,C-C CH3 H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
4 HC HC HC 0 Alcl3 H HOHC O A...View the full answer

Answered By

Kalllol Mukherjee
I am a scientist at University of Hyderabad. I have strong background in Chemistry. I have done my B.Sc and M.Sc from Visva-bharati University. I qualified NET with 70 and 34 all India rank in 2013 and 2014 respectively. I have qualified GATE in 2014 with 278 all India rank. Currently I am doing PhD in chemistry from University of Hyderabad. I have published seven research papers in internationally reputed journals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Explain the following reaction sequence. 1. Pd(OCCH,), R,P, K.CO, COCH,CH; Br 2. H,C=CH-COCH.CH,
-
Write a mechanism to explain the following reaction. (As a racemic mixture) IH2O
-
The reaction of an alkenes with bromine in an alcohol as solvent produces as ether as the product. Show a mechanism for the following reaction and explain the stereochemistry of theproduct. Br . H....
-
A small project consists of three activities: A, B, and C. To start activity C, both activities A and B need to be complete. Activity A takes 3 days with a probability of 50 percent and 5 days with a...
-
What were the primary reasons for the growth in project management during the 1990s?
-
Calculate the Fourier series coefficients C n for the function u(x) plotted as follows, but do so exploiting some of the properties contained in Eqs. (1.4)-(1.24) to provide a solution derived from...
-
Refer to the information in Exercise 24-3 and assume instead that double-declining depreciation is applied. Compute the machines payback period (ignore taxes). (Round the payback period to three...
-
The City of Green Meadows has had an employee pension fund for several years. The following is a trial balance for the fund at December 31, 2011, as well as several transactions that occurred during...
-
Assume a simultaneous open market purchase of 100 million from the Bank of England and a repayment of a discount loan of 5 million from Bank A to the Bank of England. Show the overall change in their...
-
One way to measure a persons fitness is to measure their body fat percentage. Average body fat percentages vary by age, but according to some guidelines, the normal range for men is 15-20% body fat,...
-
The 1 H NMR spectrum of the most stable isomer of [14]annulene shows two signals, at = -0.61 (4 H) and 7.88 (10 H) ppm. Two possible structures for [14]annulene are shown here. How do they differ?...
-
Metal-substituted benzenes have a long history of use in medicine. Before antibiotics were discovered, phenylarsenic derivatives were the only treatment for a number of diseases. Phenylmercury...
-
Nature's Way Inc. is planning to invest in new manufacturing equipment to make a new garden tool. The garden tool is expected to generate additional annual sales of 1,600 units at $75 each. The new...
-
Pink (Pty) Ltd is considering changing its method of inventory valuation from the absorption costing to direct costing and consulted you to determine the effect of this proposed change on the 2022...
-
Give a brief description of of lean manufacturing and costco. Briefly discuss how this trend presents challenges for Costco based on the current outlook.
-
The following data should be used for the next few questions: Raw materials inventory, January 1 $ 12,000 Raw materials inventory, December 31 16,000 Work in process, January 1 5,000 Work in process,...
-
What amount should we credit income tax payable if the tax deduction was not an aggressive position. What is income tax and gross income tax.?
-
Describe three multi domain screening instruments to be completed by professionals. Also discuss their use in detail.
-
Your client supplies water to a housing development. The company requires new customers to pay for the extension of the water lines to their homes. The water lines are the company's property. The...
-
A consultant is beginning work on three projects. The expected profits from these projects are $50,000, $72,000, and $40,000. The associated standard deviations are $10,000, $12,000, and $9,000....
-
Young's modulus for iron at room temperature is 215 GPa what strain will be produced when a mass of 10.0 kg is suspended from an iron wire of diameter 0.10 mm?
-
Poisson's ratio for lead is 0.41. What change in volume takes place when a cube of lead of volume 1.0 dm3 is subjected to an uneasily stress that produces a strain of 2.0 per cent?
-
The band gap in silicon is 1.12 eV. Calculate the minimum frequency of electromagnetic radiation that results in promotion of electrons from the valence to the conduction band.
-
The quality-control department of Starr Communications, the manufacturer of video-game DVDs, has determined from records that 1.6% of the DVDS sold have video defects, 0.9% have audio defects, and...
-
What is the half-life in hours of procainamide in a patient whose total clearance is estimated to be 20 L/h and the volume of distribution is 120 L assuming one compartment, open model, first- order...
-
1. The technologies, such as ChatGPT, has been launched before introducing even a baseline of safety measures. This goes back to trade-offs that companies make, and people just watch the impact from...

Study smarter with the SolutionInn App


