Question: When compound A (shown in the margin) is treated with dilute mineral acid, an isomerization takes place. Which of the following compounds is the new
When compound A (shown in the margin) is treated with dilute mineral acid, an isomerization takes place. Which of the following compounds is the new isomer formed?
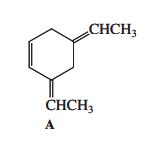
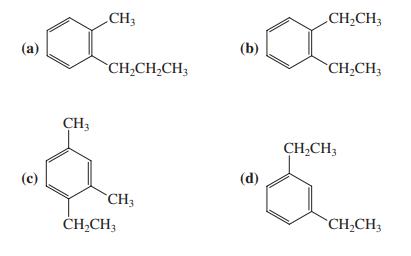
CHCH3 CHCH3 A
Step by Step Solution
3.36 Rating (183 Votes )
There are 3 Steps involved in it
CH CH3 CHCH3 d CH ... View full answer

Get step-by-step solutions from verified subject matter experts


