Ampyra is a drug used to improve mobility in patients with multiple sclerosis (MS). The skeletal structure
Question:
Ampyra is a drug used to improve mobility in patients with multiple sclerosis (MS). The skeletal structure of Ampyra is
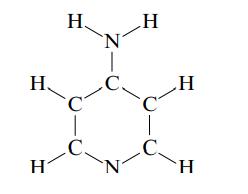
Draw the Lewis structure (including resonance forms) for Ampyra.
Transcribed Image Text:
H H H C C. N T C- N H C H H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
In the case of infrared IR visible light ultraviolet UV and ...View the full answer

Answered By

Dudhat Vaidehi
I tutored mostly elementary school students privately after school and during the summer. We met in their homes or at the public library. I charged an hourly fee, and I provided any necessary materials.
Having taught in special education in two local schools for many years meant that I had contact with a lot of parents of special needs students. I never had to advertise — word of mouth was how most folks knew of me. At one point I did have a website, but didn't utilize it much. I stayed very busy, especially in the summers, and always had a full schedule. I typically met with each student's teacher in order to get an idea of what the focus of my instruction/remediation should be. Becoming familiar with the student's learning style(s) was also very helpful. Often parents would share records and test results with me. After each tutoring session, I documented the student’s progress and gave parents written updates, as well as phone calls or emails as needed.
While my students and I certainly utilized technology and the internet often during our sessions, I never tutored online or for any tutoring company, so am not familiar with the curriculums or methods used in those settings.
Tutoring one on one was very enjoyable and rewarding. My students and I had fun, and grew quite fond of one another. The extra income was a bonus. I had to retire from tutoring due to a physically handicapping disease, and miss my students very much.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The paper Serum Beta-2-Microglobulin (SB2M) in Patients with Multiple Myeloma Treated with Alpha Interferon [Journal of Medicine (1997) 28: 311 318] reports on the influence of alpha interferon...
-
Patients with multiple sclerosis are randomly assigned a new drug or a placebo and are then given a test of coordination after six months.
-
Why do some patients with Multiple Sclerosis become unable to urinate when they have to urinate all day and night?
-
Suppose you were required to use a micrometeorite shield no more than 0.01 meters thick. What would be the required toughness of the material from which that shield was made if the shield must...
-
Liquid Gold, Inc., transports radioactive waste from nuclear power plants to disposal sites around the country. Each plant has an amount of material that must be moved each period. Each site has a...
-
What is a second mortgage?
-
Consider the following decomposition process: \[ A(\mathrm{~s}) ightleftharpoons B(\mathrm{~s})+C(\mathrm{~g}) \] The pressure at equilibrium is \(\ln P=12-\frac{3790}{T}\). Deduce the expression for...
-
Hart Lumber is considering the purchase of a paper company. Purchasing the company would require an initial investment of $300 million. Hart estimates that the paper company would provide net cash...
-
Provide a detailed explanation explaining the importance of organizational politics in the workplace. I am providing two references that you can use in your response. Please provide the pros and cons...
-
Winkler Furniture manufactures two different types of china cabinets: a French provincial model and a Danish Modern model. Each cabinet produced must go through three departments: carpentry,...
-
One type of exception to the octet rule are compounds with central atoms having fewer than eight electrons around them. BeH 2 and BH 3 are examples of this type of exception. Draw the Lewis...
-
Compare the electron affinity of fluorine to the ionization energy of sodium. Is the process of an electron being pulled from the sodium atom to the fluorine atom exothermic or endothermic? Why is...
-
In Exercises 5154, graphs of fifth-degree polynomial functions are shown. In each case, specify the number of real zeros and the number of imaginary zeros. Indicate whether there are any real zeros...
-
Some consulting work for a communications firm in Thailand. You will be paid B468,187 (Thai Baht) seven months from now when the work is done. To hedge against an unfavorable exchange rate when you...
-
Intuit's accounting solution for small businesses, startups, freelance bookkeepers, and independent accounting firms. Offering robust tools for expense tracking, tax calculation, invoice management,...
-
Describe the field and what information goes in the labeled sections: . 1. Name - 2. Category - 3. Initial quantity on hand - 4. As of Date - 5. Reorder Point - 6. Inventory Asset account 7....
-
Terry has a costume shop in a commercial building. He has a one-year lease that automatically renews for another year unless either Terry or the landlord terminates. What type of leasehold estate is...
-
in finance, searching for factors affecting stock performance is very important and potentially lucrative.An analyst decides to compare a first sample of companies with low earnings per share (EPS)...
-
Write a structural formula for each of the following compounds a. Sec-butyl tert-butyl ether b. Isoheptyl alcohol c. Sec-butylamine d. Neopentyl bromide e. 1,1-dimethylcyclohexane f....
-
In the busy port of Chennai, India, the number of containers loaded onto ships during a 15-week period is as follows: 1. Develop a linear trend equation to forecast container loadings. 2. Using the...
-
Provide the values for the missing K a or p K a in the following examples: (a) p K a = 4; K a =? (b) K a = 1 x 10 16 ; p p K a =? (c) p K a = 38; K a =? (d) K a = 1 x 10 6 ; p K a =?
-
Indicate whether these compounds are weaker or stronger acids than water (the K a for water is 1.8 x 10 ?16 ; the p K a is 15.74): (K=100) a) HCIO4 c) HOCOH (PK = 6.35) b) HCCH (pK = 25) d) CH3CH...
-
Indicate whether these species are weaker or stronger bases than hydroxide ion. The Ka is or pKa values are for the conjugate acids. a) :NH, (K = 10-38) c) NH, (PK, = 9.24) b) CHCHCH d) :CI: (pK =...
-
A 2-meter-tall basketball player attempts a goal 12.4 meters from the basket that is 3.05 meters high. If he shoots the ball at a 55 angle, at what initial speed must he throw the basketball so that...
-
Find the following. 2. 3 y(t) + 3y(t) 3+ = x(t) + 3 x(t) 3. y(t) - 1/2 y(t) x(t) = dt d 4. dt d 5. dt y(t) + 1/2 y(t) + y(t) y(t) + 1/2 y(t) = x(t) + x(t) = x(t)
-
1. Who are the people affected by rsum truth decisions? 2. Why might a job seeker have a duty to blur parts of his or her work history? 3. Why might an egoist lie on the rsum, and why not? 4. Does an...

Study smarter with the SolutionInn App


