Consider the following half-reactions: A hydrochloric acid solution contains platinum, palladium, and iridium as chloro-complex ions. The
Question:
Consider the following half-reactions:
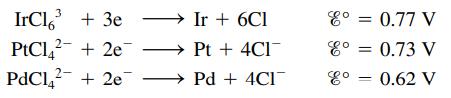
A hydrochloric acid solution contains platinum, palladium, and iridium as chloro-complex ions. The solution is a constant 1.0 M in chloride ion and 0.020 M in each complex ion. Is it feasible to separate the three metals from this solution by electrolysis? (Assume that 99% of a metal must be plated out before another metal begins to plate out.)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:





