Name the compounds in parts ad and write the formulas for the compounds in parts eh. a.
Question:
Name the compounds in parts a–d and write the formulas for the compounds in parts e–h.
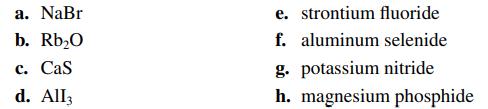
Transcribed Image Text:
a. NaBr b. Rb₂0 c. Cas d. All3 e. strontium fluoride f. aluminum selenide g. potassium nitride h. magnesium phosphide
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
a NaBr Sodium bromide b RbO Rubidium oxi...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Write formulas for and name the binary hydrogen compounds of the second-period elements (Li to F). Describe how the physical and chemical properties of these compounds change from left to right...
-
Write expanded formulas for the following compounds and name them using the IUPAC system: a. (CH3)3CCH2CH2CH3 b. CH3(CH2)2CH3 c. (CH3)2CHCH2CH2CH3 d. CH3CCl2CF3 e. (CH2)4 f. CH3CH2CHFCH3 g. EtBr h....
-
Write the molecular and structural formulas for the compounds represented by the following molecular models: (a) (b) (c) (d) P F F F
-
1. If net profit is $47,025; depreciation is $2,200; accounts receivable increases $5,056; accounts payable increases $4,673; and inventory declines $850; what is the operating cash flow for the...
-
Pamello, Inc., an engineering consulting firm, uses the cash method of accounting and is a calendar year taxpayer. Compute the amount of Pamellos current year deductions for the following...
-
If you're swimming in the ocean near the coast of North Korea right now, then you're not doing logic homework on an electronic device. You are doing logic homework on an electronic device. Therefore,...
-
A uniform bar of length \(l\) and mass \(m\) is hinged at one end \((x=0)\), supported by a spring at \(x=\frac{2 l}{3}\), and acted upon by a force at \(x=l\), as shown in Fig. 13.32. Derive the...
-
Marinette Company makes several products, including canoes. The company has been experiencing losses from its canoe segment and is considering dropping that product line. The following information is...
-
Watch the video An Insider's Plan for Rehabilitating the Juvenile Justice System, Jeff Wallace ( https://www.youtube.com/watch?v=TOxpjjzP6lM ). and use Shawn's case chosen from the PBS cases Four...
-
The finance director of RM plc is considering several investment projects and has collected the following information about them. Projects D and E are mutually exclusive. The capital available for...
-
Complete the following table. Atom/lon 50 Sn 25Mg+ 2+ 56 Fe+ 120S 79 32 Se 34- 35C1 53 Cu Protons Neutrons Electrons
-
For carbon-14 and carbon-12, how many protons and neutrons are in each nucleus? Assuming neutral atoms, how many electrons are present in an atom of carbon-14 and in an atom of carbon-12?
-
What is the energy of a beam of electrons that exhibits a first-order maximum at an angle of when diffracted by a crystal grating with a lattice plane spacing of 0.215 nm?
-
Internal Evaluation. Internal analysis of the company, to include the analysis of: The company's current situation and the key challenges it needs to overcome (if any) - any existing purpose...
-
How to modify the following algorithms to display the total number of swap operations. The following algorithms are given: def Algorithm_1(lyst): i = 0 while i < len(lyst) - 1: minIndex = i j = i + 1...
-
Propose a NEW operating systems. The NEW operating systems can be based on Windows or Unix/Linux or Mobile OS or independent based platform. Provide details and justifications of your NEW operating...
-
1) What is a material handling system? Explain. 2) List and describe the 5 uses of packaging. 3) Advise on some ways that a company can minimize its potential cargo loss. 4) List 5 types of commonly...
-
DBS Bank is launching a new investment product targeting senior citizens in Asia. The company decides to use direct marketing for selling the product. 1. What direct marketing strategy would allow...
-
Lerner Corporation wholesales repair products to equipment manufacturers. On April 1, 2016, Lerner Corporation issued $12,000,000 of five-year, 8% bonds at a market (effective) interest rate of 6%,...
-
The following data are supplied for the common stocks of Nikola Corporation, Tesla, Inc. and General Motors: Nikola Corp (NKLA) Tesla Inc. (TSLA) Close Price ($) Close Price ($) 67.53 30.00 40.81...
-
The 1980s saw reports of fH (SiH2) ranging from 243 to 289 k] mol-1. For example, the lower value was cited in the review article by R. Walsh (Ace. Chem. Res. 14,246 (1981)); Walsh later leant...
-
Given that tG = -212.7 kJ mol-1 for the reaction in the Daniell cell at 25C, and b (CuS04) = 1.0 x 10-3 mol kg-i and b (ZnS04) = 3.0 x 10-3 mol kg-I, calculate (a) The ionic strengths of the...
-
Although the hydrogen electrode may be conceptually the simplest electrode and is the basis for our reference state of electrical potential in electrochemical systems, it is cumbersome to use....
-
Given the function. f(x) = ln (2x+3x) Compute f'(x). Enter your answer rounded to the nearest tenth
-
A motor-cycle manufacturing company desires a profit of $600,000. The fixed costs are $800,000. The unit selling price of the motor-cycle is $750 and the variable cost per unit is $680. How many...
-
3. ZR Corporation's stock has a beta coefficient equal to 0.8 and a required rate of return equal to 11 percent. If the expected rate of return on the market is 12.5 percent, what is the risk-free...

Study smarter with the SolutionInn App


