One method for determining the purity of aspirin (C 9 H 8 O 4 ) is to
Question:
One method for determining the purity of aspirin (C9H8O4) is to hydrolyze it with NaOH solution and then to titrate the remaining NaOH. The reaction of aspirin with NaOH is as follows:
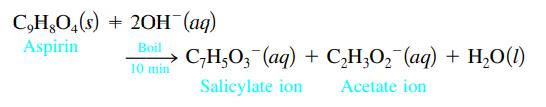
A sample of aspirin with a mass of 1.427 g was boiled in 50.00 mL of 0.500 M NaOH. After the solution was cooled, it took 31.92 mL of 0.289 M HCl to titrate the excess NaOH. Calculate the purity of the aspirin. What indicator should be used for this titration? Why?
Transcribed Image Text:
C,H,O4(s) + 2OH(aq) Aspirin Boil C₂H5O3(aq) + C₂H₂O₂ (aq) + H₂O(1) 10 min Salicylate ion Acetate ion
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
Solution a The purity of the aspirin NaOH x ml NaOH used to titrate excess mL HCl used 0500 M x ...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
What is the equation method for determining the break-even point? Explain how the results of this method differ from those of the contribution margin approach.
-
Determining Diet One method for determining the amount of com in early Native American diets is the stable isotope ratio analysis (SIRA) technique. As com photosynthesizes, it concentrates the...
-
One method for determining the position of an image, either real or virtual, is by means of parallax. If a finger or other object is placed at the position of the image, as shown in Figure Q36.25,...
-
The Best Buy Co. Inc. 10-K report has the following footnote related to leasing activities. The future minimum lease payments under our capital and operating leases by fiscal year (not includ- ing...
-
Has the availability of shelf registrations reduced the importance of private placements? Why?
-
Walters Audio Visual, Inc., offers a stock option plan to its regional managers. On January 1, 2021, 40 million options were granted for 40 million $1 par common shares. The exercise price is the...
-
True or False. The Newmark method is an implicit integration method.
-
a. Find the weekly diet that meets the nutritional requirements in the least costly manner. What is the lowest possible minimum cost? What preference rating does this solution have? b. Find the...
-
Flint Distribution markets CDs of numerous performing artists. At the beginning of March, Flint had in beginning inventory 2,700 CDs with a unit cost of $6. During March, Flint made the following...
-
Piedmont Manufacturing produces metal products with the following standard quantity and cost information: Overhead rates were based on normal monthly capacity of 6,000 machine hours. During November,...
-
What quantity (moles) of HCl(g) must be added to 1.0 L of 2.0 M NaOH to achieve a pH of 0.00? (Neglect any volume changes.)
-
You make 1.00 L of a buffered solution (pH = 4.00) by mixing acetic acid and sodium acetate. You have 1.00 M solutions of each component of the buffered solution. What volume of each solution do you...
-
The year-end Trial Balance of a business owned by Stella Hanson included the ledger balances listed on the right. Stella wishes to report the land and buildings at their current market value on 30...
-
A client has completed a loan application. All documents are in order and she meets the lender's criteria to be approved for the loan. However, something just does not feel right to you. You feel...
-
Appraise Bank of America's existing risks for the previous five years. Evaluate the strengths and weaknesses of the three main risk transfer methods - hedging, insuring, and diversifying,...
-
Luciana passed away at the age of 75 with $50,000 remaining in her RRIF. She named her grandson Sebastian, who is 16 as beneficiary. Sebastian's family is interested in helping him save on the taxes...
-
You have 2 sisters, Chrystia and Melanie. Your sisters are 32 years old. Your sisters wish to retire when they are 57 years old. Each of your sisters expect to live for 30 years after they retire....
-
How do they make money (list all the sources)- In terms everyone in class can understand? What are their key ratios: Pick 3 of the following and describe what the ratio is in terms everyone can...
-
A 15-A circuit breaker trips repeatedly. Explain why it would be dangerous to replace it with a 20-A circuit breaker.
-
Reduction in sales All of the above 29. Belt of an electric motor is broken, it needs a. Corrective maintenance b. Scheduled maintenance c. Preventive maintenance d. Timely maintenance. 30. The...
-
When acetic acid is treated with isotopically labeled water ( 18 O, shown in red) in the presence of a catalytic amount of acid, it is observed that the isotopic label becomes incorporated at both...
-
Phosgene is highly toxic and was used as a chemical weapon in World War I. It is also a synthetic precursor used in the production of many plastics. (a) When vapors of phosgene are inhaled, the...
-
Fluphenazine is an antipsychotic drug that is administered as an ester prodrug via intramuscular injection: The hydrophobic tail of the ester is deliberately designed to enable a slow release of the...
-
A scuba diver is exploring a shipwreck at a depth of 30 meters in the ocean. The diver's tank contains 5.0 moles of compressed air at a temperature of 25C. What is the pressure of the air in the tank...
-
1. Determine the atmospheric pressure at a location where the barometric reading is 720 mm Hg. 2. A pressure gauge connected to a tank reads 52 psi at a location where the barometric reading is 29.6...
-
Assume that a bond has an annual coupon rate of 6.80 percent, but makes coupon payments on a semi-annual basis, has a par value of $1,000, a yield to maturity of 7.20 percent, and a current price of...

Study smarter with the SolutionInn App


