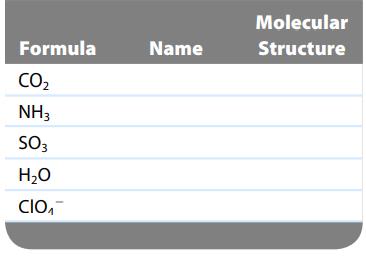
The formulas of several chemical substances are given in the table below. For each substance in the
Question:
The formulas of several chemical substances are given in the table below. For each substance in the table, give its chemical name and predict its molecular structure.
Transcribed Image Text:
Formula CO₂ NH3 SO3 H₂O CIO₁ Name Molecular Structure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
CO2 Carbon Dioxide OCO N...View the full answer

Answered By

Hande Dereli
Enthusiastic tutor, skilled in ACT and SAT tutoring. Raised one student's score on the SATs from 1100 combined to 1400. Graduated with a 3.9 GPA from Davidson College and led a popular peer tutoring group for three years. Scored in the top 0.06% in the nation on the SATs. The real reason I'm the one to help you nail the test? Results. Clients invariably praise my ability to listen and communicate in a low-stress, fun way. I think it's that great interaction that lets me raise retest SAT scores an average of 300 points.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The data given in the table below are the midterm scores in a course for a sample of 10 students and the scores of student evaluations of the instructor. (In the instructor evaluation scores, 1 is...
-
The descriptive statistics shown in the table below for the variable Income from the US Incomes data set shows that it is negatively skewed? Income (Per Year) Mean 30598.28712 Standard Error...
-
a. Fill the blanks in the table below for a quarterly comparison of sectors with the S&P 500 Index (see the box on page 581). b. Did the portfolio manger under- or over perform the S&P 500? c. Which...
-
Izmir A.S. issued convertible bonds at their face value of 100,000 lira on December 31, 2020. The bonds have a 10-year life with interest of 10 percent payable annually. At the date of issue, the...
-
Jason Wright is a part time business student who would like to optimize his financial decisions. Currently, he has $ 16,000 in his savings account. Based on an analysis of his take home pay, expected...
-
Listed below are selling prices (dollars) of TVs that are 60 inches or larger and rated as a "best buy" by Consumer Reports magazine. Are the resulting statistics representative of the population of...
-
For each of the following situations, calculate the degrees of freedom \((d f)\), identify the critical values (assume \(\mathrm{a}=.05\) [two-tailed]), calculate the \(t\)-statistic \((t)\), make a...
-
Income statements for Thompson Company for 2011 and 2012 follow. Required a. Perform a horizontal analysis, showing the percentage change in each income statement component between 2011 and 2012. b....
-
a. Performed $8,200 of services on account. b. Collected $5,600 cash on accounts receivable. c. Paid $1,450 cash in advance for an insurance policy. d. Paid $400 on accounts payable. e. Recorded the...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Which of the following compounds or ions exhibit resonance? a. O 3 b. CNO - c. AsI 3 d. CO 3 2- e. AsF 3
-
Look up the energies for the bonds in CO and N 2 . Although the bond in CO is stronger, CO is considerably more reactive than N 2 . Give a possible explanation.
-
Use rules of exponents to simplify the expression. Use positive exponents to write your answer. 1-41-x9 9x-2y3
-
What is responsibility accounting? Discuss its significance in divisional performance measurement.
-
Variances are not an end in themselves. In the light of the above statement, explain briefly what you understand by variances control and responsibility?
-
Fill in the following blanks. In some cases choices are provided. (i) Responsibility accounting focuses on ______________________. (ii) _______________is a sub-unit of an organisation under the...
-
Why is ROI classified as a composite performance measure? Explain its significance.
-
What are the two approaches to disposing off variances? Which will be preferred, when, and why? Take suitable examples to support your viewpoint.
-
Section 6.3.4 discusses 802.11 mobility, in which a wireless station moves from one BSS to another within the same subnet. When the APs are interconnected with a switch, an AP may need to send a...
-
A manufacturer can sell product 1 at a profit of $20 per unit and product 2 at a profit of $40 per unit. Three units of raw material are needed to manufacture one unit of product 1, and six units of...
-
Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of each designated bond and the orbital's that are overlapping to form it? (both) TTT a) HC=C_C_CH ...
-
What is the hybridization at all atoms, except hydrogen's, in these compounds? a) H H H H H H H H CH 4.6 c) HC-N-CH3 : : g) CHC-OH h) CHC-NHCH,
-
Draw the p orbital's that compose the conjugated part of these molecules: a) :-CH3 b) CH,=CHNH, c) H-C-C-CH=CH
-
Anna has an investment that will bring her $100 with a 30% probability and $40 with a 70% probability. Anna's Utility function is U = Y (1/2) . Where Y= income. Anna is considering selling this...
-
on January 1, 2000, the price of koka kola was $10. on jan and, 2020, the shares were worth $100. the stock Paid no dividends during the period. what is the annual geometric return.
-
Explain why leasing is an option for a company expansion. include, what leasing is and how it will benefit the company in it's expanding efforts. Also, how is capital or operating leasing recorded on...

Study smarter with the SolutionInn App


