The rate constant for the gas-phase decomposition of N 2 O 5 , has the following temperature
Question:
The rate constant for the gas-phase decomposition of N2O5,
![]()
has the following temperature dependence:
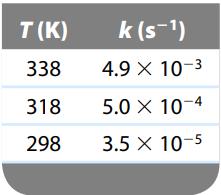
Make the appropriate graph using these data, and determine the activation energy for this reaction.
Transcribed Image Text:
N₂O, (g) 2NO₂(g) + O₂(g)
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The temperature dependence of the rate constant for a reaction is tabulated as follows: Temperature (K) k (M-1 s-1) 600................................0.028 650................................0.22...
-
The rate constant for the gas- phase decomposition of N2O5, N2O5 2NO2 + 1/2O2 has the following temperature dependence: Make the appropriate graph using these data, and deter-mine the activation...
-
A) For the gas phase decomposition of hydrogen iodide at 700 K 2 HIH2 + I2 the average rate of disappearance of HI over the time period from t = 0 s to t = 1446 s is found to be 5.6710-4 M s-1. The...
-
Suppose that, in an attempt to raise more revenue, Nowhere State University (NSU) increases its tuition. Will this necessarily result in more revenue? Under what conditions will revenue (a) rise, (b)...
-
What is viral marketing? How do marketers use brand ambassador or brand evangelists?
-
Sketch an Engel curve corresponding to Figure 4.12 in Economics, 10th edition. What can we say about the income elasticity of demand for an inferior good?
-
Plaintiff visited South Chicago on January 10, 2008, seeking a new 2008 Nissan Versa (Versa) with manual transmission, anti-lock brakes, and other features. He was told by the employees of South...
-
Apple Inc., headquartered in Cupertino, California, designs, manufactures, and markets mobile communication and media devices, personal computers, and portable digital music players and sells a...
-
On January 1, 2023, Blossom Ltd. acquires a building at a cost of $200,000. The building is expected to have a 20-year life and no residual value. The asset is accounted for under the revaluation...
-
3. Andi and Brandon contribute to a public good they both enjoy. Let a denote a contribution by Andi, b a contribution by Brandon. Andi and Brandon may choose any nonnegative contribution levels....
-
DDT (molar mass = 354.49 g/mol) was a widely used insecticide that was banned from use in the United States in 1973. This ban was brought about due to the persistence of DDT in many different...
-
One reason suggested for the instability of long chains of silicon atoms is that the decomposition involves the transition state shown below: The activation energy for such a process is 210 kJ/mol,...
-
At the end of the current year, $22,650 of fees have been earned but have not been billed to clients. a. Journalize the adjusting entry to record the accrued fees. b. If the cash basis rather than...
-
If a company wishes to make a change in accounting principle in an interim period, but is not able to determine the effects of the change on the current fiscal year's previous interim periods, the...
-
How long is the etchant material applied to the tooth being prepared for use of a bonding material? Group of answer choices 5 to 10 seconds 15 to 20 seconds 1 minute It is left on the tooth and is...
-
Vehicle operating costs incurred for the month totaled $7,840. Total miles drive were 22,400, distributed as follows: Construction by Work Orders $11,200 Retirement by Work Orders $1,120 Operation of...
-
Under IFRS, long-term investments in bonds must ANSWER Unselected use the effective interest rate method. Unselected use either the straight-line or the effective-interest rate method. Unselected use...
-
An effective progress note records specific types of information and excludes other types of information. Select the correct types of information that should be included in an effective progress...
-
Find the maximum value and the minimum value of the function and the values of x and y for which they occur. a. P = 17x - 3y + 60, subject to 6x + 8y 48, 0 y 4, 0 x 7 b. Q = 28x - 4y + 72,...
-
CRUZ, INC. Comparative Balance Sheets December 31, 2015 CRUZ, INC. Income Statement For Year Ended December 31, 2015 Required Use the indirect method to prepare the cash provided or used from...
-
Compounds A and B are isomers with the formula C3H6O. A has a peak at 1730 cm ?1 in its IR spectrum and B has a peak at 1715cm ?1 , the mass spectra of A and B are as follows, show the structures of...
-
Compounds C and D are isomers with the formula C9H12, in addition to other absorption peaks, both compounds show a peak near 7.25 ? (area 5) in their 1H-NMR spectra. Their mass spectra are as follow,...
-
Compounds E and F are isomers with the formula C6H8 both react with H2 in the presence of Pt to give G (C6H12), G shows a single peak in its 13C-NMR spectrum. E has no absorption maximum above 200nm...
-
Solve the following complex number problems. Solutions for complex number transformation is required in this item. a. Simplify: 120 +31 18-2115 2111 +5 b. (32-120)+(1/120) (4-1)(-1-1) 5e C. 2e0.25mi...
-
(a) Let3+ and 2 = a + bi be complex numbers. Suppose that 7 Argument = 12' find Argument(22). 7-2 (b) Let the map f: CC be defined by f(z) = Find f() if=1+2i. 1 (c) Solve the equation -12 i(9-2),...
-
3. Suppose f: RR is continuous and U CR. Assume R has the usual Euclidean metric. (a) Define U is "open" in R.

Study smarter with the SolutionInn App


