Use the Lewis acidbase model to explain the following reaction. CO(g) + HO(1) HCO3(aq)
Question:
Use the Lewis acid–base model to explain the following reaction.
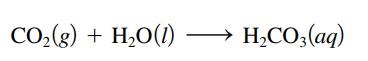
Transcribed Image Text:
CO₂(g) + H₂O(1) H₂CO3(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
The Lewis acidbase model explains chemical reactions in terms of the transfer of electrons from a Le...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Propose a mechanism to explain the following reaction by using words, and in doing so predict the absolute stereochemistry (R or S) at the stereocentre in the product. Then draw three other product...
-
Use Lewis symbols to show the reaction of atoms to form arsine, AsH3. Indicate which electron pairs in the Lewis formula of AsH3 are bonding and which are lone pairs.
-
The following are representations of acid base reactions: a. Label each of the species in both equations as an acid or a base and explain. b. For those species that are acids, which labels apply:...
-
At year-end 2010, 28,879 million represents: A. the funded status of the plan. B. the defined benefit obligation. C. the fair value of the plans assets. Kensington plc, a hypothetical company based...
-
Explain the four major principles of risk classification.
-
An idealized radial turbine is shown in Fig. P11.87. The absolute flow enters at 30° and leaves radially inward. The flow rate is 3.5 m3/s of water at 20°C. The blade thickness is constant at...
-
True or False: A bearing that shows signs of brinelling but no sign of surface deformation or corrosion can yield a full life.
-
Briefly describe FASB/IASB convergence process and the principles that guide their convergence efforts.
-
Evaluate the challenges and strategies associated with implementing quality control in complex, global supply chains, considering the impact of multi-tier suppliers, geographical dispersion, and...
-
A physics student pulls a block of mass m = 22 kg up an incline at a slow constant velocity for a distance of d = 4.5 m. Does the incline make an angle? = 32? with the horizontal. The coefficient of...
-
Is an aqueous solution of NaHSO 4 acidic, basic, or neutral? What reaction occurs with water? Calculate the pH of a 0.10-M solution of NaHSO 4 .
-
Calculate the pH of a 0.010-M solution of iodic acid (HIO 3 , K a = 0.17).
-
Given the number 10 8 , ____________ is the base.
-
BuyCo, Inc., holds 29 percent of the outstanding shares of Marqueen Company and appropriately applies the equity method of accounting. Excess cost amortization (related to a patent) associated with...
-
Change the total fixed manufacturing overhead cost for the Milling Department in Data area back to $390,000, keeping all of the other data the same as in the original example. Consider a new job, Job...
-
Tom selis mutual funds on a graduated commission structure. He receives 3.1% on the first $40,000 of sales in a month 4.2% on the next $40,000, and 5.3% on all further sales. What are his gross...
-
A hamburger factory produces 55,000 hamburgers each week. The equipment used costs $15,000 and will remain productive for three years. The labor cost per year is $14,500. a. What is the productivity...
-
Matthew, Inc., owns 30 percent of the outstanding stock of Lindman Company and has the ability to significantly influence the investee's operations and decision making. On January 1, 2021, the...
-
If objects of weights x and w1 are suspended from the balance in Figure 13(A), the cross-beam is horizontal if bx = aw1. If the lengths a and b are known, we may use this equation to determine an...
-
Using Apple, demonstrate how the differentiation strategy can be well implemented.
-
The hydrolysis of a biological thioester to the corresponding carboxylate is often more complex than the overall result might suggest. The conversion of succinyl CoA to succinate in the citric add...
-
One step in the gluconeogenesis pathway for the biosynthesis of glucose is the partial reduction of 3-phosphoglycerate to give glyceraldehydes 3-phosphate. The process occurs by phosphorylation with...
-
Penicillins and other fl-lactam antibiotics (see the Focus On in this chapter) typically develop a resistance to bacteria due to bacterial synthesis of -lactamase enzymes. Tazobactam, however, is...
-
Pharoah Enterprises purchased a delivery truck on January 1 , 2 0 2 5 , at a cost of $ 2 6 , 0 0 0 . The truck has a useful life of 7 years with an estimated salvage value of $ 5 , 9 1 0 . The...
-
Consider the following recurrence relation: A(1)=1, A(n) = 2A(n-1)+2"-1 Use the method of unraveling to find a closed form for A(n).
-
Given the following formula: Monthly Pay= [rate + rate / ([1+rate] ^(months) -1) X principle Where rate of 6% means 6/1200 and Months means number of years x 12 Possible data to test: Principle 12200...

Study smarter with the SolutionInn App


