What amount (moles) of compound is present in 1.00 g of each of the compounds in Exercise
Question:
What amount (moles) of compound is present in 1.00 g of each of the compounds in Exercise 53?
Data in Exercise 53?
Calculate the molar mass of the following substances.
a. 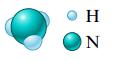
b. 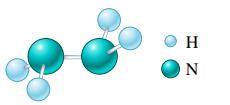
c. (NH4)2Cr2O7
Transcribed Image Text:
Η N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
c NH42Cr2O7 To calculate the molar mass of NH42Cr2O7 we need to sum up the atomic mas...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Calculate the molar mass of the following substances: (a) Li2 CO3, (b) CS2, (c) CHCl3 (chloroform), (d) C6H8O6 (ascorbic acid, or vitamin C), (e) KNO3, (f) Mg3N2.
-
What mass of compound is present in 5.00 moles of each of the compounds in Exercise 54? Data In Exercise 54? Calculate the molar mass of the following substances. a. b. Ca 3 (PO 4 ) 2 c. Na 2 HPO 4 0
-
What mass of compound is present in 5.00 moles of each of the compounds in Exercise 53? Data in Exercise 53? Calculate the molar mass of the following substances. a. b. c. (NH 4 ) 2 Cr 2 O 7 OH N
-
Find the x-values where the following do not have derivatives. + -6 + y 8 0 6 X
-
Use the Holt Winters no trend model to find the best model to find forecasts for the next 12 months in the Excel file Housing Starts.
-
Paula is planning to either purchase or lease a $75,000 automobile. She anticipates that business use of the auto will be 60% for the first two years but will decline to 40% in years three through...
-
What is cryptojacking? How can you protect yourself from being a victim of cryptojacking?
-
Emarpy Appliance produces all kinds of major appliances. Richard Feehan, the president of Emarpy, is concerned about the production policy for the company's best selling refrigerator. The demand for...
-
Suppose your firm receives a $4.01 million order on the last day of the year. You fill the order with $1.76 million worth of inventory. The customer picks up the entire order the same day and pays...
-
PROJECT STEPS 1. Gudrun Duplessis is a manager in the Alanis Parks Department. Gudrun is compiling spending data in preparation for a city bond offering for park funding. She wants to use Excel to...
-
What is the mass of the product? a. Less than 10 g b. Between 20 and 100 g c. Between 100 and 120 g d. Exactly 120 g e. More than 120 g
-
Fig. 3.5 illustrates a schematic diagram of a combustion device used to analyze organic compounds. Given that a certain amount of a compound containing carbon, hydrogen, and oxygen is combusted in...
-
An analysis of the income statement and the balance sheet accounts of Holmes Export Co. at December 31 of the current year, provides the following information. Additional Information 1. Except as...
-
B2 ) Consider the following information on a portfolio of three stocks: State of Economy Probability of State of Economy Boom .15 Stock A Rate of Return .05 Stock B. Rate of Return 21 Stock C Rate of...
-
14. Consider the following information on three stocks: Probability of State Rate of Return if State Occurs State of Economy of Economy Stock A Stock B Stock C Boom 0.45 0.55 0.35 0.65 Normal 0.50...
-
Part 1 - Rent Calculation Assume the following for a floor in a multistory office building: total usable area: 62,500 sq ft; Total common area: 4,100 sq ft; total rentable area: 66,600 sq ft; Tenant...
-
Sport L&K Company produces annual cash flows of $748 dollars and is expected to exist forever. (Numbers are in thousands). The company is currently financed with 59 percent equity. Your analysis...
-
8) The data below relates to the accounts of a firm which manufacturers a single product. Factory wages, 60% of which are paid to production staff 340,000 Rent & rates, 85% of which is for the...
-
a. What is the hybridization of each of the carbon atoms in the following compound? b. What is the hybridization of each of the carbon, oxygen, and nitrogen atoms in the following compounds? CH CHCH...
-
Which task is performed by a book-keeper? A. Analysing the trading results B. Entering transactions in the ledger C. Preparing year-end financial statements D. Providing information for...
-
This hydroboration reaction forms two products. Show these products and explain which one you expect to be amajor. 1) BH3, THF 2) H,O2. NAOH
-
Show preparation of these alcohols fromalkenes. b) c)
-
Show the products of thesereactions: 1) BH3, THF 2) H2O2, NaOH a) Ph- b) 1) disiamylborane 2) HO2, NaOH
-
Jackson, a self-employed taxpayer, uses his automobile 90% for business and during 2023 drove a total of 15,000 business miles. Information regarding his car expenses is listed below. Business...
-
Bel Air Flying Service started operations on December 1 , 2 0 . The trial balance in Figure 1 3 . 6 was prepared at month - end. Record the following adjustments in the General Journal, then make an...
-
Founded in 1960, the Arpeggio Music Company is a family-owned and operated firm specializing in the educational music market. Located in the U.S. Middle West, its products and services include...

Study smarter with the SolutionInn App


