Determine the mass of CO 2 produced (kg) per unit of energy released (kJ), i.e., the CO
Question:
Determine the mass of CO2 produced (kg) per unit of energy released (kJ), i.e., the CO2 emission, for the complete combustion of the following fuels. The higher heating value (HHV) expresses the energy released per mass of fuel burned.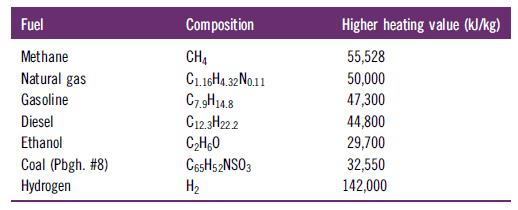
Transcribed Image Text:
Fuel Methane Natural gas Gasoline Diesel Ethanol Coal (Pbgh. #8) Hydrogen Composition CH4 C1.16H4.32 No.11 C7.9H14.8 C12.3H222 C₂H60 C65H52NSO3 H₂ Higher heating value (kJ/kg) 55,528 50,000 47,300 44,800 29,700 32,550 142,000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
To calculate the CO2 emission factor we need to use the equation CO2 emission factor kg CO2kg fuel m...View the full answer

Answered By

Diana Muriuki
As an online math tutor, I have several years of hands-on experience working with students of all ages and skill levels. I hold a Bachelor's degree in Mathematics and a Master's degree in Education. Additionally, I have completed multiple training courses in online teaching and tutoring methods.
Throughout my career, I have worked with students in both individual and group settings, including classroom teaching, after-school tutoring, and online instruction. I am proficient in teaching a wide range of math topics, from basic arithmetic to advanced calculus and statistics.
One of my greatest strengths as a tutor is my ability to adapt my teaching style to meet the unique needs and learning styles of each individual student. I understand that every student is different, and I strive to create a comfortable and supportive learning environment that encourages growth and development.
In addition to my formal education and tutoring experience, I am also a lifelong learner with a passion for mathematics. I am constantly seeking out new resources and methods to improve my own knowledge and skills, and I believe this passion and enthusiasm helps to inspire my students as well.
Overall, my hands-on experience and proficiency as a math tutor are grounded in a combination of formal education, practical experience, and a genuine love of mathematics. I am confident in my ability to help students achieve their goals and succeed in math, and I look forward to the opportunity to work with new students and continue to grow as an educator.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
In Problem 12.54, you calculated the CO 2 emission factor for several hydrocarbon fuels. Derive an expression for the CO 2 emission factor for an arbitrary hydrocarbon C x H y in which the...
-
Methanol (CH3OH) is used as a fuel in race cars. (a) Write a balanced equation for the combustion of liquid methanol in air. (b) Calculate the standard enthalpy change for the reaction, assuming...
-
Ethanol (C2H5OH) is currently blended with gasoline as an automobile fuel. (a) Write a balanced equation for the combustion of liquid ethanol in air. (b) Calculate the standard enthalpy change for...
-
If you were a hedge fund manager, which style would you employ and why? Describe this style and how it works. Why would you utilize it and under what macroeconomic conditions would this style work...
-
ExxonMobil repurchased common shares during 2011 and, as a result, the Treasury Stock account increased by $20,324 million. The market price of ExxonMobil shares averaged $81.50 per share during the...
-
LSUS Consulting provides 3 months consulting service for 6,000 to Scheinert Inc. starting Jan 1, 2023. Please assume service are performed evenly throughout months. On Jan 31, 2023 LSUS prepared a...
-
(i) Describe the construction and working of a spark plug. Where it is used ? Why? (ii) An engine working on Otto cycle, air has a pressure of \(1 \mathrm{bar}\) and temperature of \(27^{\circ}...
-
Pantene Paints has annual sales of $ 2,250,000 with variable expenses of 60 percent of sales and fixed expenses per month of $ 25,000. By how much must annual sales increase for Pantene Paints to...
-
Identify the reasons for disclosure below as they are (OR) are not for public interest or benefit. Options 1. Not for public interest or benefit 2. Not for public interest or benefit 3. For public...
-
The box plot below shows the amount spent for books and supplies per year by students at four year public colleges. a. Estimate the median amount spent. b. Estimate the first and third quartiles for...
-
Determine the total standardized enthalpy H (kJ) for a fuelair reactant mixture containing 1 kmol CH 4 , 2.5 kmol O 2 , and 9.4 kmol N 2 at 500 K and 1 atm. Also determine the mass-specific...
-
Determine the standardized enthalpies of the following pure species at 4 atm and 2500 K: H 2 , H 2 O, and OH.
-
In the text, we ignored travelers checks in deriving the money multiplier. Suppose we are more careful and include travelers checks in the money supply. Let T be the level of travelers checks, so T/D...
-
Read the following and answer the questions below. The costs of hosting the Olympic Games During Olympic years there is a tangible sense of anticipation for many people who enjoy watching or...
-
If an employer requests a rsum, should you also include a cover letter even though the employer didnt specifically ask for one? Explain.
-
Research a geographic area where you would like to work. Investigate the cost of living, industrial growth in the area, weather and climate, and attractions in the area you could visit. The local...
-
Living in Pickering, Lauren Bossers worked virtually by e-mail and phone for a supply chain management software company in Winnipeg. Even though Bossers was a remote worker, she was shocked when her...
-
The best way to conduct performance evaluation is to determine broad business goals and prepare a financial plan in which senior management is responsible for the whole business. Discuss.
-
Explain what is meant by recalculation and reperformance. Give an example of each type of audit evidence. Why are recalculation and reperformance often dual-purpose tests?
-
On October 1, 2021, Adoll Company acquired 2,600 shares of its $1 par value stock for $38 per share and held these shares in treasury. On March 1, 2023, Adoll resold all the treasury shares for $34...
-
A manufacturer of cutting tools has developed two empirical equations for tool life in hours (y 1 ) and for tool cost in dollars (y 2 ). Both models are linear functions of steel hardness (x 1 ) and...
-
A central composite design is run in a chemical vapor deposition process, resulting in the experimental data shown on the next page. Four experimental units were processed simultaneously on each run...
-
Verify that an orthogonal first-order design is also first-order rotatable.
-
Estimate the Q, W, and change in the enthalpy, entropy, and internal energy for the following case: Liquid A with mass = 1 kg Initial volume 2195 cm/kg The liquid is compressed at constant...
-
Katrina owns undeveloped land with an adjusted basis of $300,000. She exchanges it for other undeveloped land worth $750,000. Assume that Katrina holds the land as an investment. If an amount is...
-
If Canada did not buy and sell goods internationally, how would your community be affected?

Study smarter with the SolutionInn App


