To establish the positivity of the specific heat at constant pressure Cp compressibility coefficient at constant temperature
Question:
To establish the positivity of the specific heat at constant pressure Cp compressibility coefficient at constant temperature κT (see relations (6.31)), follow the steps given here [65]:a) Show that the Mayer relation (5.42) can be recast as,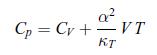 where α is the thermal coefficient of expansion,
where α is the thermal coefficient of expansion,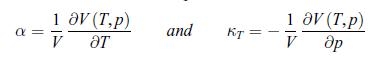
b) Show that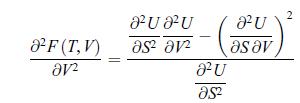
c) Conclude from these two results that κT ≥ 0 and Cp ≥ 0.
Transcribed Image Text:
Cp=Cv+VT КТ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
To establish the positivity of the specific heat at constant pressure Cp and the compressibility coe...View the full answer

Answered By

Ragul r
I am highly experienced and dedicated educator with a passion for teaching students of all ages. With over
4 years of experience in education and tutoring, I have helped countless students achieve their academic goals in subjects such as math and science.
My journey in education began with a Bachelor's degree in from Maseno University, where I developed a strong foundation in critical thinking and problem-solving skills that have proved invaluable in my teaching career.
I am committed in helping your students in IT and various topics in sciences and math
0.00
0 Reviews
10+ Question Solved
Related Book For 

Principles Of Thermodynamics
ISBN: 9781108426091
1st Edition
Authors: Jean-Philippe Ansermet, Sylvain D. Brechet
Question Posted:
Students also viewed these Engineering questions
-
The specific heat at constant pressure for an ideal gas is given by cp = 0.9 + (2.7 x 10-4) T (kJ/kg K) where T is in kelvin. The change in the enthalpy for this ideal gas undergoing a process in...
-
The specific enthalpy (h) of an ideal gas is a function of temperature (T) only as can be seen from Table D-3 for air. (a) Using the data from the table, determine the specific heat at constant...
-
The specific heat at constant pressure cp [J/(kg K)] of an ideal gas is related to enthalpy by where h = enthalpy (kJ/kg), and T = absolute temperature (K). The following enthalpies are provided for...
-
Question 4: Artistic Hand for engineering Services was formed on Jun 1, 2018. The following transactions took place during the month of Jun: I The owner invested $20,000 cash in business. 2 Hired an...
-
The following events occurred as part of the operations of Craig State University, a public university: 1. To construct a new computer complex, the university floated at par a $22,000,000, 7% serial...
-
1. A spaceship is heading directly toward Earth at a velocity v of 0.720c. The astronaut on board claims that he can send a canister toward the Earth at u = 1.30c relative to Earth. Calculate the...
-
Internal auditors are often used to review an organizations financial statements such as balance sheets, income statements, and cash flow statements prior to public filings. Auditors seek to verify...
-
Cars arrive at Carla's Muffler shop for repair work at an average of 3 per hour, following an exponential distribution. (a) What is the expected time between arrivals? (b) What is the variance of the...
-
You are working as an analyst for a potential investor in a new wind farm. The wind farm will be located in a small electricity market. The market currently has one major generation firm and several...
-
You have just been hired by Internal Business Machines Corporation (IBM) in their capital budgeting division. Your first assignment is to determine the free cash flows and NPV of a proposed new type...
-
An isolated system consisting of two closed subsystems A and B is separated by a diathermal wall. Initially, they are held at temperatures TiA and TiB . Subsystem A contains NA moles of gas. The...
-
A system consists of a thin film of surface area A, of internal energy U(S, A), where Hence, the surface tension is given by Express the heat Qif to provide to the film for a variationAif = AfAi of...
-
Stratton County Community College is adding a band program that will be open to college students and also to the community. The music department chair has asked you to prepare a special- purpose...
-
The required rate of return for Air Asia. a. Use the CAPM method to determine rs. b. Use the Treasury 10-year bond as the rRF. Month Dec-20 Nov-20 Oct-20 Sep-20 Aug-20 Jul-20 Jun-20 Hay-20 Apr-20...
-
Investor Dan has $390,000 to invest in bonds. Bond A yields an average of 6% and the bond B yields 9.6%. Dan requires that at least 4 times as much money be invested in bond A as in bond B. You must...
-
Rewrite following code by directly setting the corresponding registers using minimum number of lines of code. setup(){ pinMode(7.INPUT); pinMode(10,OUTPUT); pinMode(11,INPUT_PULLUP);...
-
As part of a weight reduction program, a man designs a monthly exercise program consisting of bicycling, jogging, and swimming. He would like to exercise at most 40 hours, devote at most 8 hours to...
-
24. Both you and your older brother would like to have $28,500 in 14 in years. Because of your success in this class, you feel that you are a more savvy investor than your brother and will be able to...
-
If you create a regression model for estimating the Height of a pine tree (in feet) based on the Circumference of its trunk (in inches), is the slops most likely to be 0.1, 1, 10, or 100? Explain.
-
One study found that the elderly who do not have children dissave at about the same rate as the elderly who do have children. What might this finding imply about the reason the elderly do not dissave...
-
In an adiabatic constant-volume container, a fluid is to be stirred mechanically with a motor providing 40 W of shaft power. How long can this go on if the internal energy of the fluid is not allowed...
-
Calculate the density of gaseous H 2 O at each of the following states:a. 0.07 bar, 360 K,b. 1 atm, 700 K,c. 200 bar, 700 K,Using the perfect gas model. Compare the values obtained with the values...
-
The temperature of 10 kg of air inside a pistoncylinder is increased from 25C to 80C using an electrical heater, while the pressure inside the cylinder is maintained constant. During this process 50...
-
Larsen and Buss present a simple formula for calculating heritability. One simply takes two times the difference between the correlation ("r") on some trait for monozygotic (MZ) or identical twins...
-
Sally loves her new Aqualand swimming pool. Unfortunately, the pool ladder is too high so if someone slides down head first they will likely receive injuries to head and their neck and in some cases,...
-
Mr Johnathon B. Good is the owner of two new Range Rover Sport Utility Vehicles. He decided to sell the black 2022 model to pay for a new apartment. He put a "for sale" sign on the Range Rover. Mr...

Study smarter with the SolutionInn App


