The reaction of a compound with silver nitrate in ethanol is used as a chemical test to
Question:
The reaction of a compound with silver nitrate in ethanol is used as a chemical test to determine if the compound is an alkyl halide. The formation of a precipitate of the silver halide constitutes a positive test.
(a) Explain why these conditions favor the SN1 mechanism.
(b) Which of these halides would give a precipitate more rapidly when reacted with AgNO3 in ethanol?
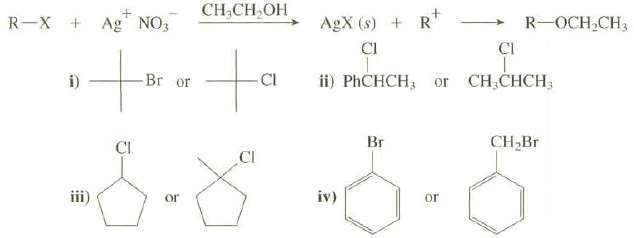
Transcribed Image Text:
AgX (s) + R* CI CH,CH,OH R-X + Ag NO3 R-OCH CH3 CI -Br or CI ii) PHCHCH; or CH,CHCH3 i) Br CH,Br Cl CI iii) or or iv)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
a The solvent ethanol is polar and there are no strong nucleophiles pr...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Determine the structure of a compound with molecular formula C 5 H 10 O that exhibits the following broadband-decoupled and DEPT-135 spectra. The DEPT-90 spectrum has no signals. Broadband-decoupled...
-
The IR spectrum of a compound with molecular formula C5H8O was obtained in CCl4 and is shown in Figure 13.42. Identify the compound. Wavelenga qum) 15 16 14 3600 340) 3800 3300 3000 280K 2600 2400...
-
A ketone can be prepared from the reaction of a nitrile with a Grignard reagent. Describe the intermediate that is formed in this reaction, and explain how it can be converted to a ketone.
-
Scandinavo Ltd. is a CCPC that began operations on January 1, 2020 when it was first incorporated and a calendar fiscal period was chosen. Scandinavo Ltd. Is not associated with any other...
-
Woodchuck Timber Pty Ltd grows, harvests and processes timber for use in the building industry. The following data relate to the company's sawmill during June: Work in process, 1 June: Direct...
-
Mark and Pamela are equal partners in MP Partnership. The partnership, Mark, and Pamela are calendar year taxpayers. The partnership incurred the following items in the current year: a. Compute the...
-
Air enters a frictionless, constant area duct with \(\mathrm{Ma}=2.5\), \(T_{0}=20^{\circ} \mathrm{C}\), and \(p_{0}=101 \mathrm{kPa}(\mathrm{abs})\). The gas is decelerated by heating until a normal...
-
During company estimates that total factory overhead costs will be $750,000 for the year. Direct labor hours are estimated to be 300,000. For Darling company, (a) Determine the predetermined factory...
-
A scooter has a 3.5 litres petrol tank. It uses 15 ml of petrol per lap of a 300m track. a) If Sarah fills up the tank, how many laps of the track can she do? b) How many kilometres can Sarah travel...
-
The following are data regarding last years production of Dicer Ricer, one of the major products of Kitchen Gadget Company. During the year, 61,000 units of this product were manufactured and 62,100...
-
Ethers can be cleaved by treatment with strong acids. Show all of the steps in the mechanism for this reaction and explain why these products are formed rather than iodomethane and2-methyl-2-butanol:...
-
The reaction of an alkyl chloride (or bromide) with sodium iodide in acetone proceeds according to the following equation: Sodium iodide is soluble in acetone, whereas both sodium chloride and sodium...
-
A business using the retail method of inventory costing determines that merchandise inventory at retail is $396,400. If the ratio of cost to retail price is 61%, what is the amount of inventory to be...
-
Day trading refers to buying and selling securities (stocks and bonds, for example) in the same day, with hopes of an upward price movement. What information does a day trader need to make accurate...
-
There are over twenty thousand objects orbiting in space. For a given object, let \(A\) be the event that the charred remains do hit the earth. Suppose experts, using their knowledge of the size and...
-
Comarketing and cobranding are techniques that companies often use to market their own and each other's products, such as Sherwin Williams paints featured in Pottery Barn home furnishings catalogs....
-
With reference to Exercise 4.87, find the mean and the variance of the distribution of the number of microelectrodes made from glass tubing using (a) the probabilities obtained in that exercise; (b)...
-
No matter how powerful they are, comic book heroes can't get themselves into bookstoresand readers' handswithout a little help. Leon Avelino and Barry Matthews, co-founders of Secret Acres, know that...
-
A single-phase, three-wire panelboard must feed 30 circuits. From tables provided in this chapter, identify the minimum frame size required. TABLE 18.4 COMMON PANELBOARD AND LOAD CENTER RATINGS AND...
-
Using the theoretical sampling strategy, how many samples of size 4 (n = 4) can be drawn from a population of size: (a) N = 5? (b) N = 8? (c) N = 16? (d) N = 50?
-
Sketch the graph of each line. y=5 --4-3-2-1 15 4 3 19 44 61 1 2 3 to 56x
-
Treatments of a 1, 3-diketone such as 2,4-pentanedione with base does not give an aldol condensation product. Explain.
-
What product would expect to obtain from base treatment of 1,6-cyclo-decacedione? Base 1,6-Cyclodecanedione
-
Show the products you would except to obtain by claisen condensation of the following esters: (a) (CH3)2CHCH2COEt (b) Ethyl phenyl acetate (c) Ethyl cyclohexylacetate
-
On January 1, 2023, Bertrand, Incorporated, paid $86,900 for a 40 percent interest in Chestnut Corporation's common stock. This investee had assets with a book value of $229,500 and liabilities of...
-
Landen Corporation uses a job-order costing system. At the beginning of the year, the company made the following estimates: Direct labor-hours required to support estimated production Machine-hours...
-
Explain the concept of gene-environment interactions, and how these interactions influence the expression of complex traits and the risk of developing multifactorial diseases ?

Study smarter with the SolutionInn App


