Question: Use Table 4.2 to predict whether the equilibrium for these reactions favors the reactants or the products. a) :NH + CHCCH, NH + CHCCH, O
Use Table 4.2 to predict whether the equilibrium for these reactions favors the reactants or the products.
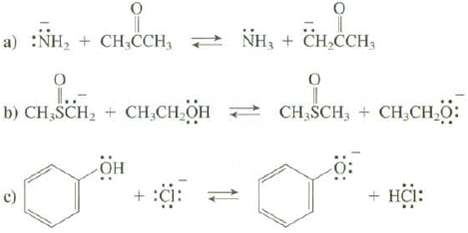
a) :NH + CHCCH, NH + CHCCH, O 1..- b) CHSCH + CHCHOH + :C: 9 CH SCH, CHCHO: o + HCI:
Step by Step Solution
3.51 Rating (161 Votes )
There are 3 Steps involved in it
Remember the equilibrium favors the formation of the weaker acid and the weaker base The ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-A-B-R (21).docx
120 KBs Word File


